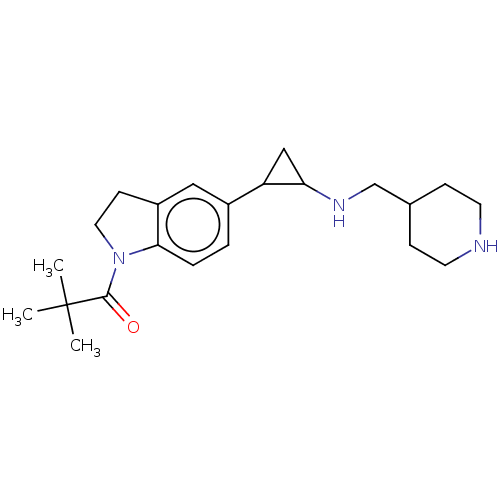
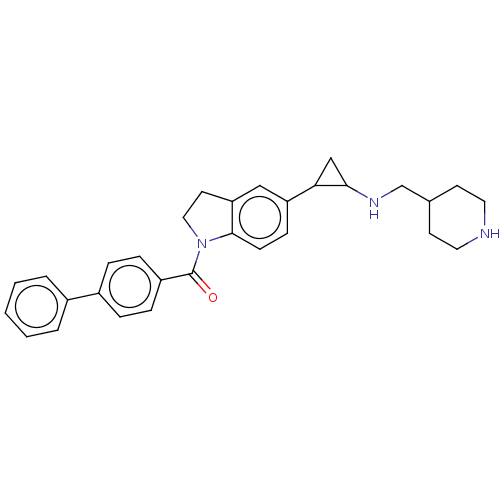
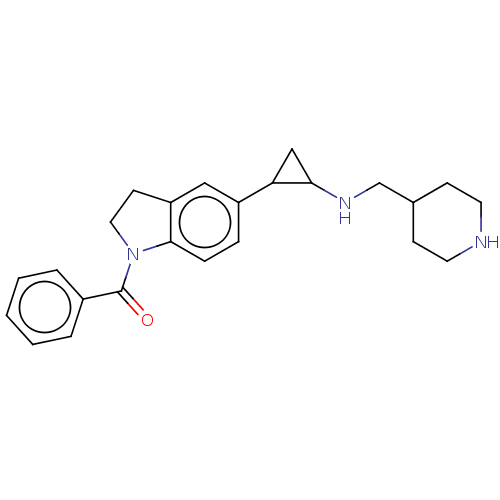
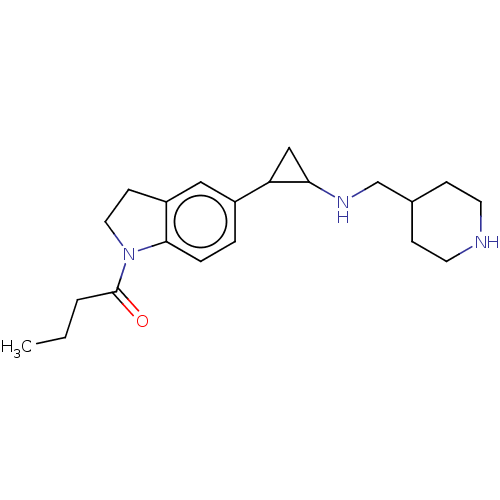
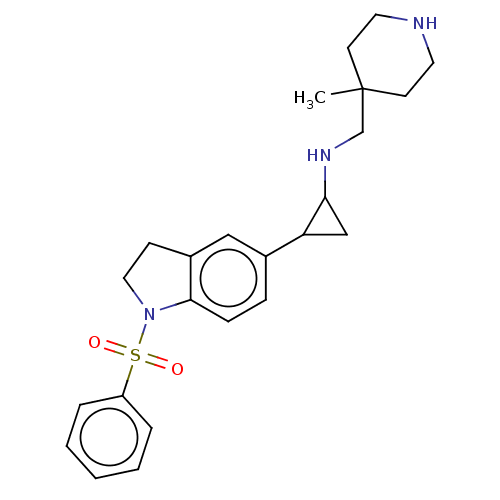
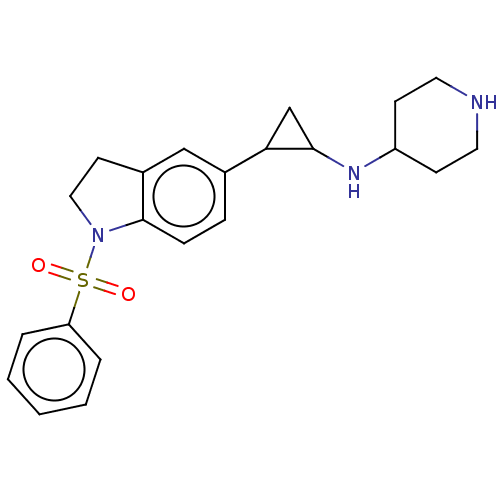
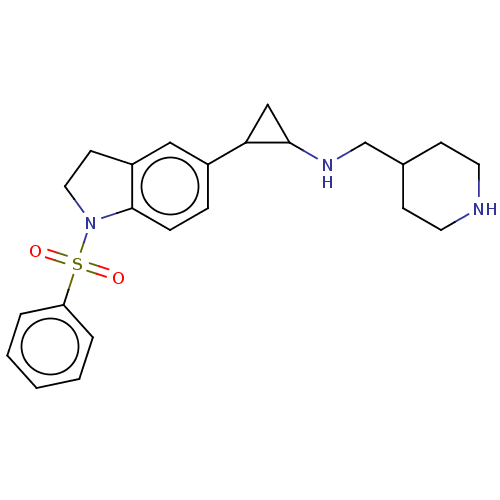
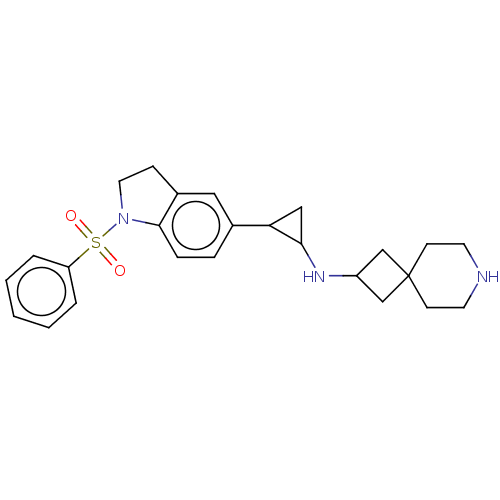
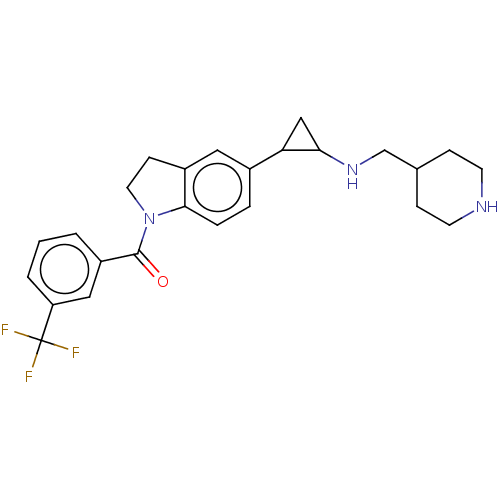
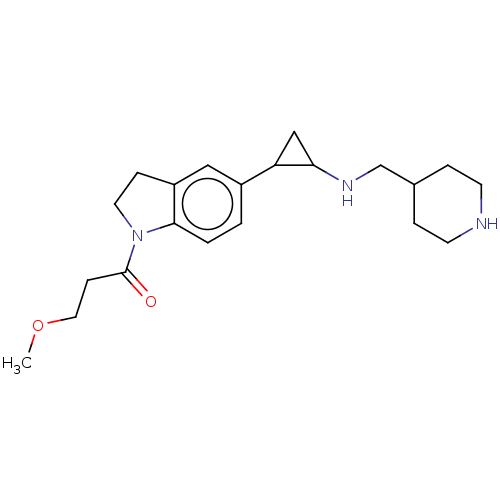
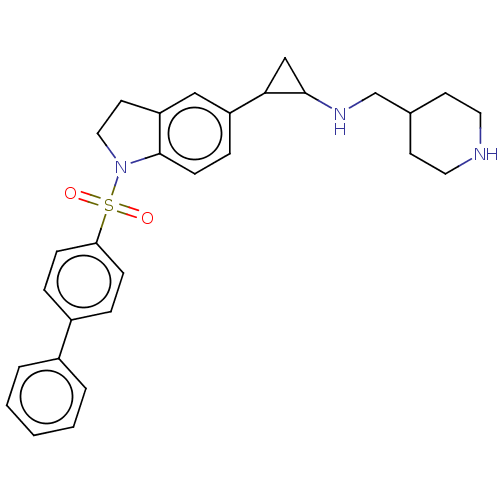
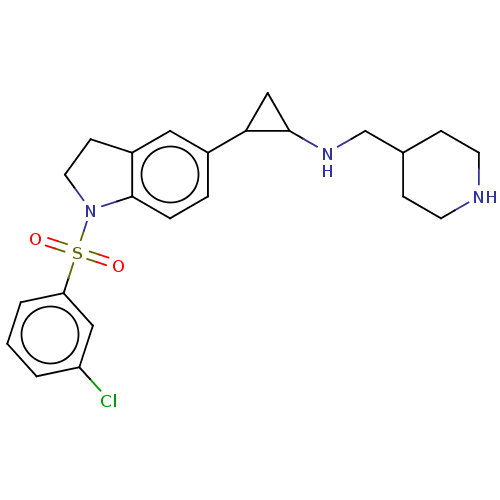
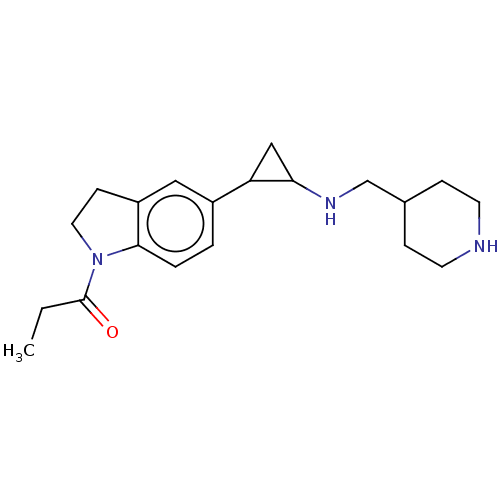
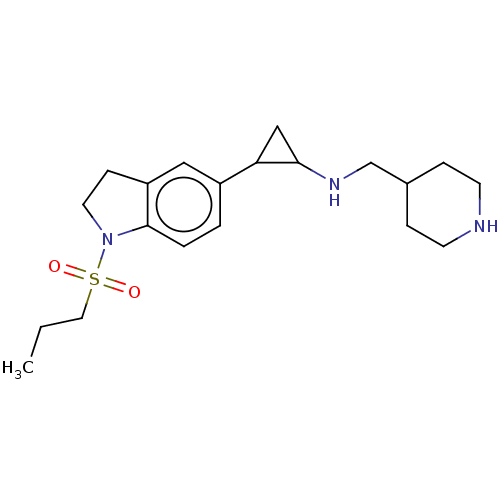
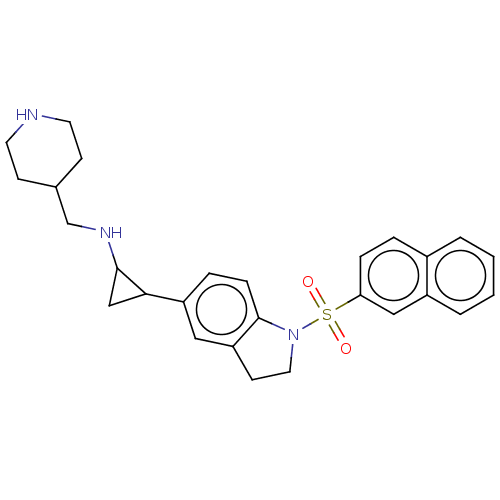
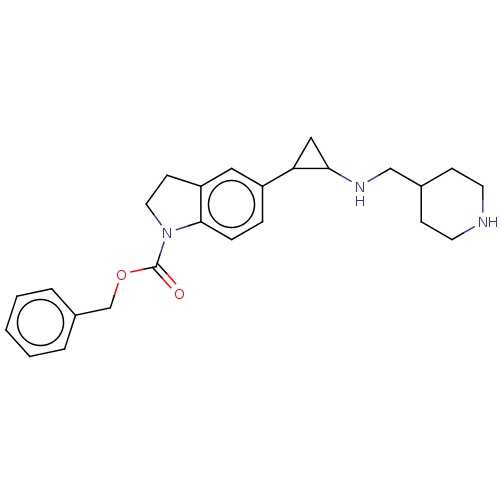
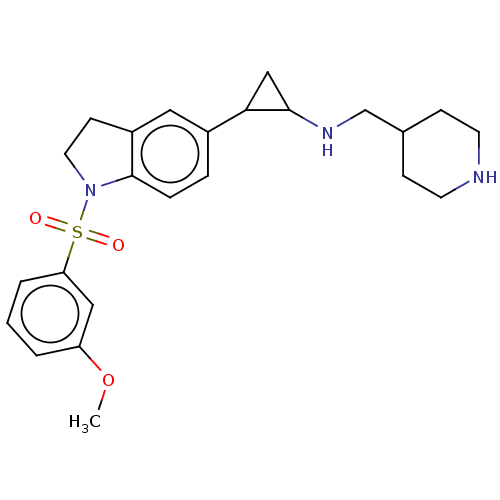
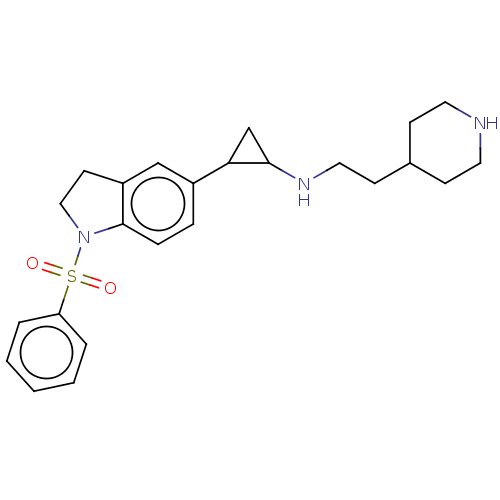
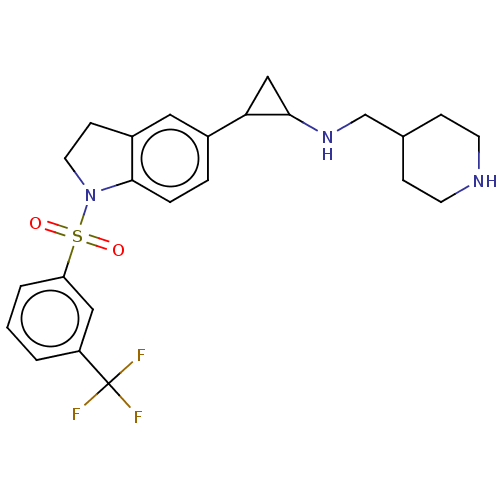
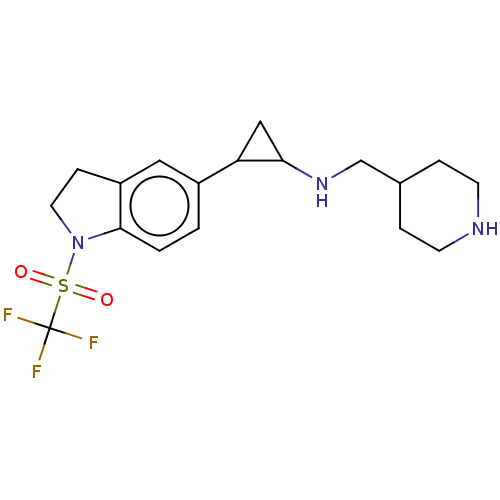
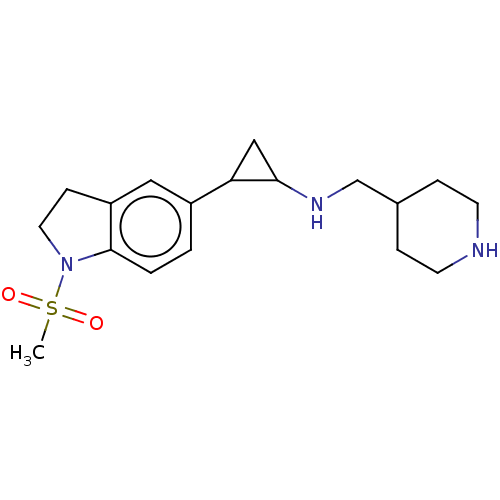
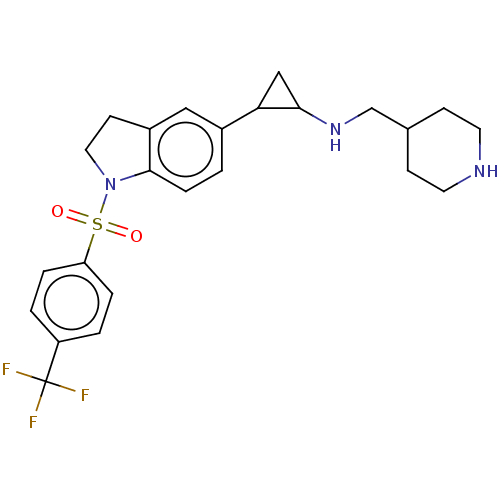
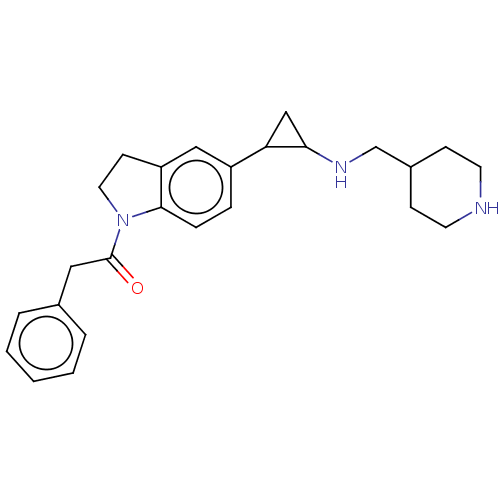
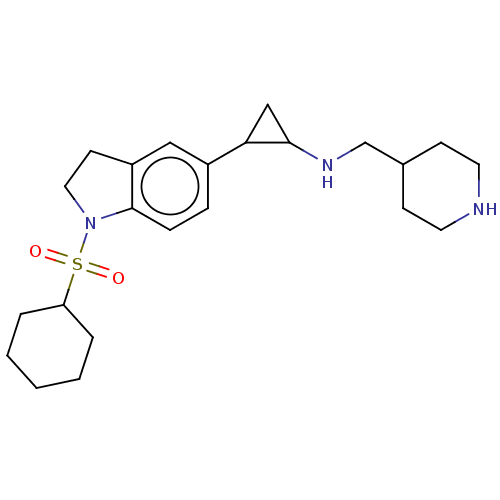
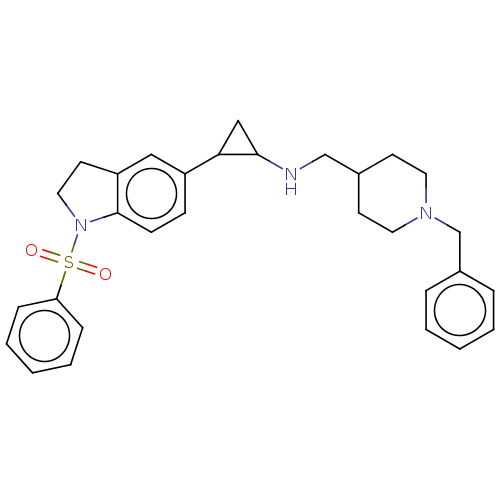
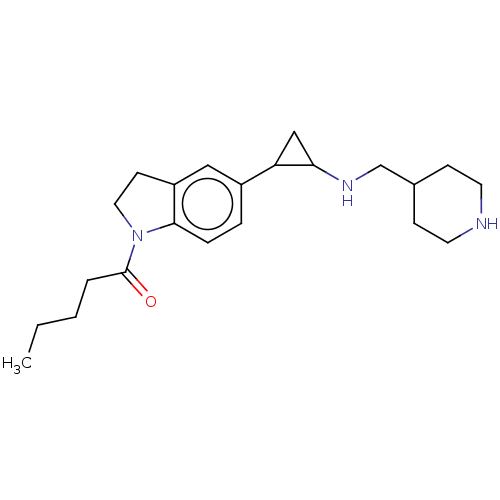
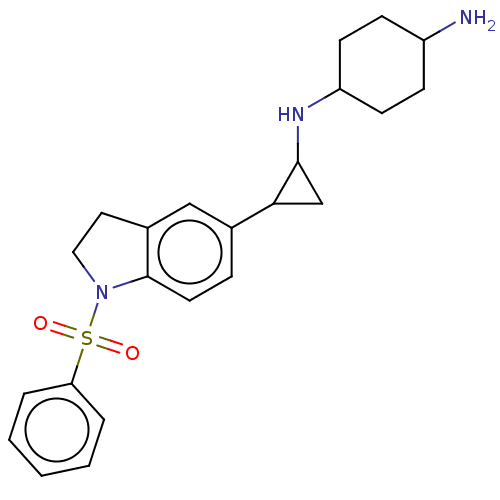
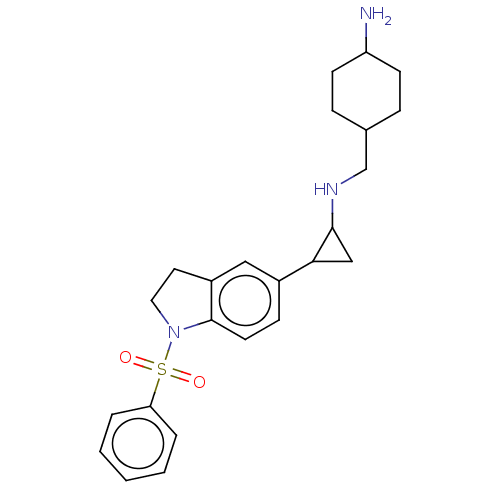
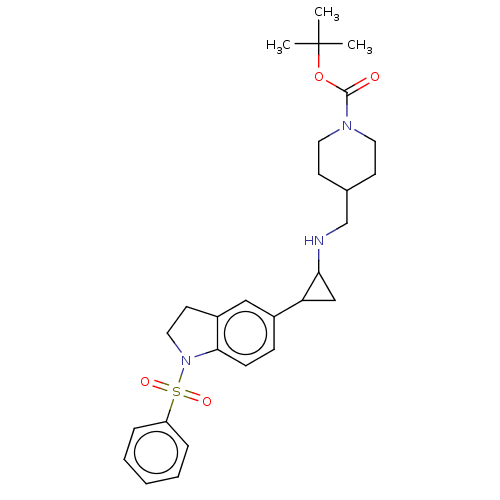
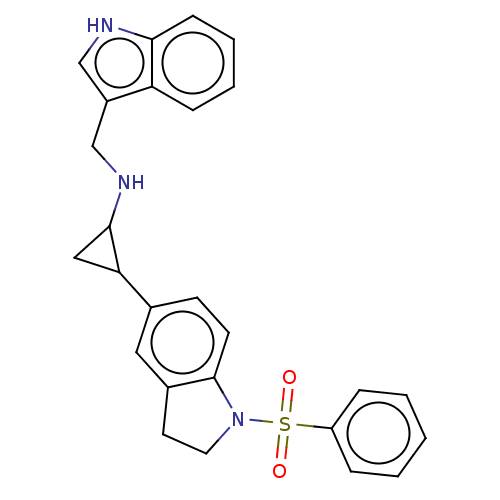
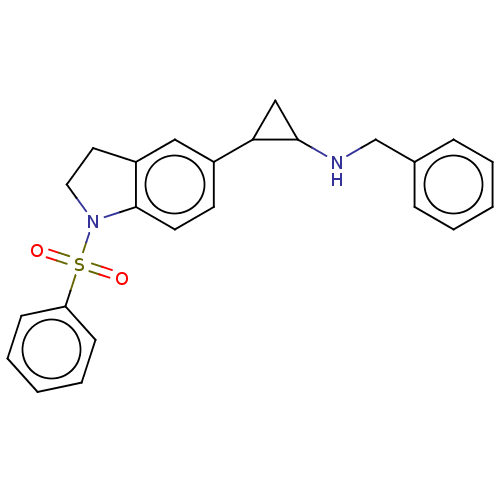
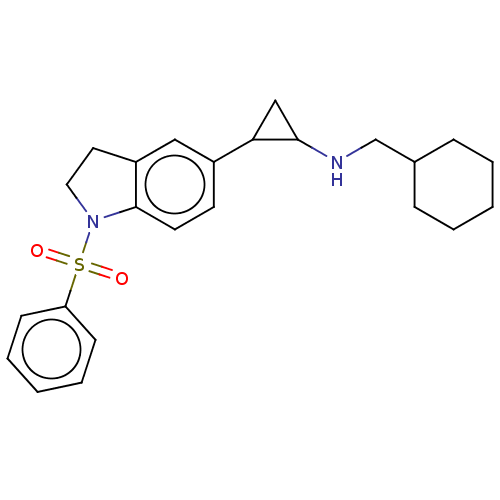
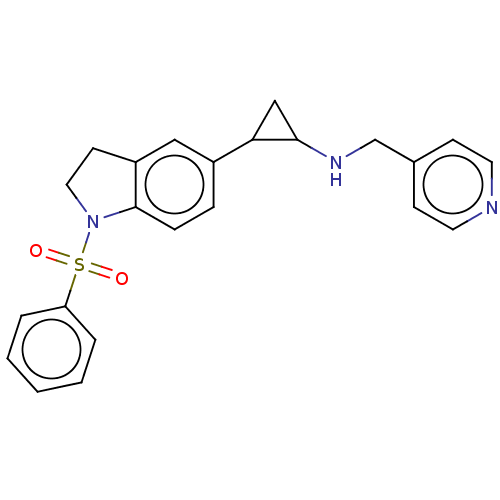
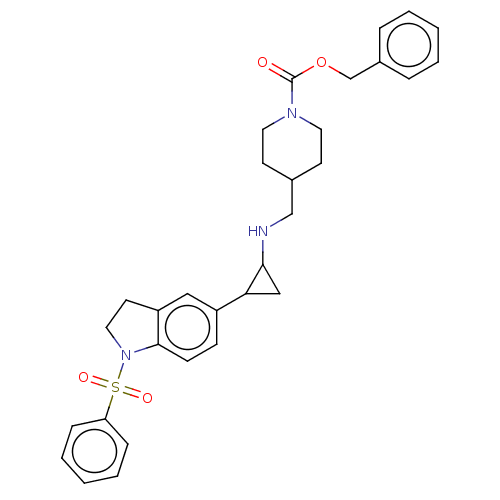
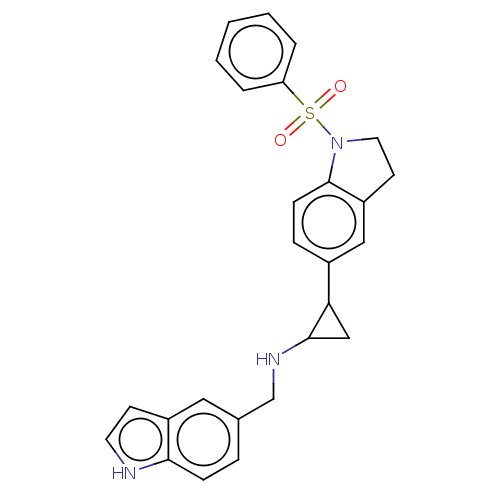
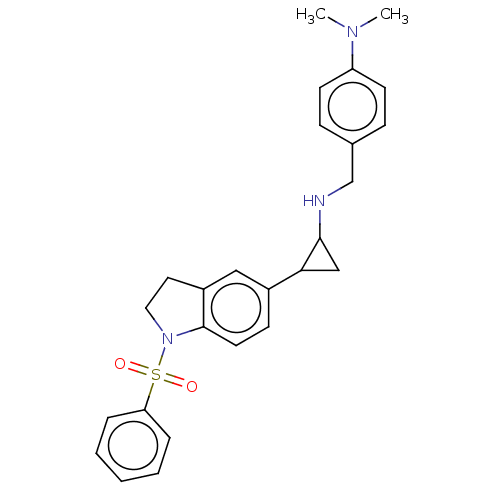
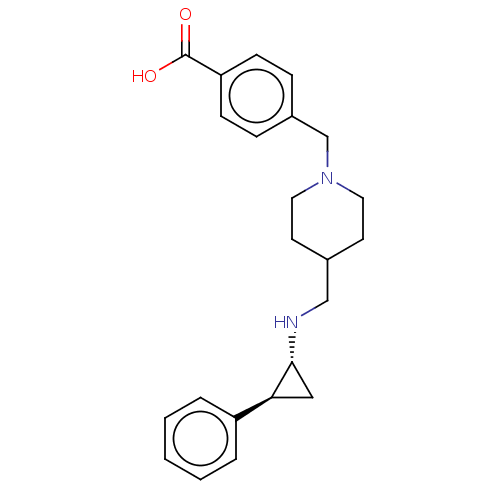
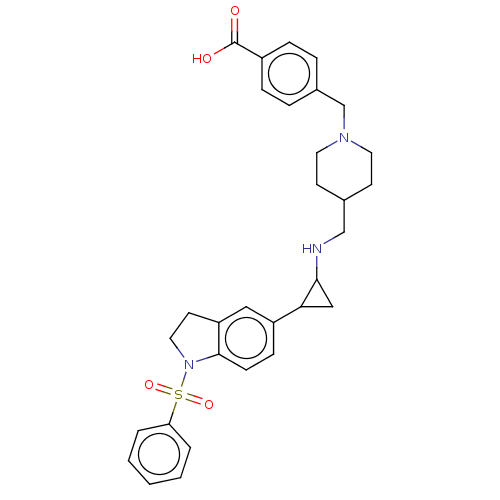
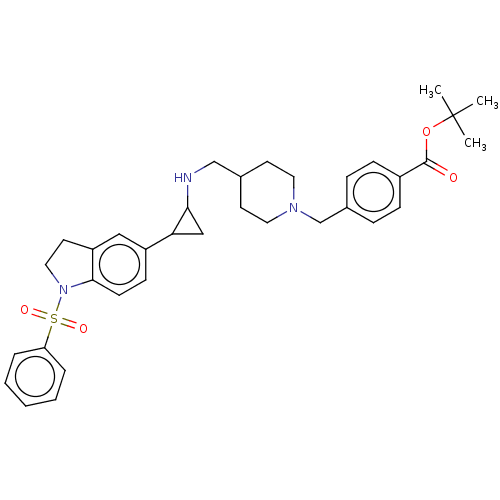
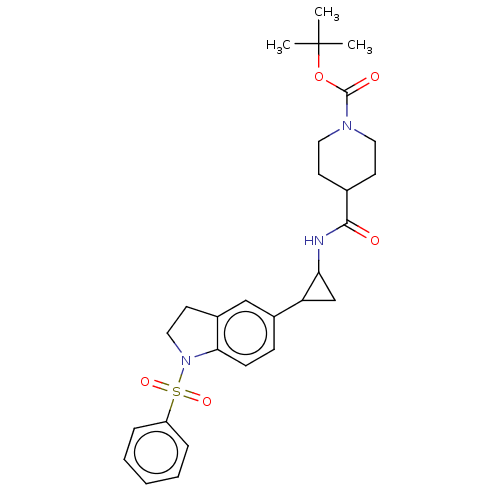
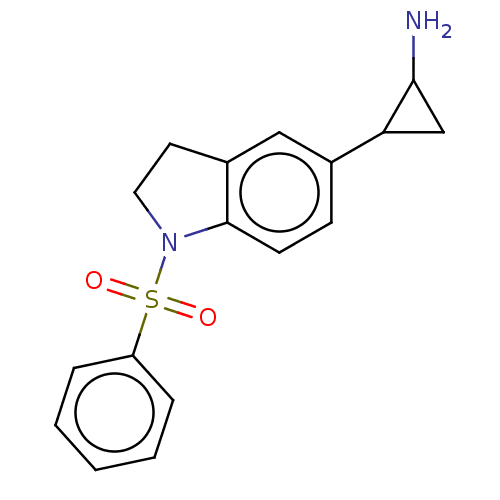
Report error Found 106 Enz. Inhib. hit(s) with all data for entry = 11760
Affinity DataIC50: 8.72nMAssay Description:Principle: LSD1 specifically removes the methylation modification at K4 lysine on H3 polypeptide substrate, making it a substrate without methylation...More data for this Ligand-Target Pair
Affinity DataIC50: 10.2nMAssay Description:Principle: LSD1 specifically removes the methylation modification at K4 lysine on H3 polypeptide substrate, making it a substrate without methylation...More data for this Ligand-Target Pair
Affinity DataIC50: 15.2nMAssay Description:Principle: LSD1 specifically removes the methylation modification at K4 lysine on H3 polypeptide substrate, making it a substrate without methylation...More data for this Ligand-Target Pair
Affinity DataIC50: 15.6nMAssay Description:Principle: LSD1 specifically removes the methylation modification at K4 lysine on H3 polypeptide substrate, making it a substrate without methylation...More data for this Ligand-Target Pair
Affinity DataIC50: 17.1nMAssay Description:Principle: LSD1 specifically removes the methylation modification at K4 lysine on H3 polypeptide substrate, making it a substrate without methylation...More data for this Ligand-Target Pair
Affinity DataIC50: 22.8nMAssay Description:Principle: LSD1 specifically removes the methylation modification at K4 lysine on H3 polypeptide substrate, making it a substrate without methylation...More data for this Ligand-Target Pair
Affinity DataIC50: 24.4nMAssay Description:Principle: LSD1 specifically removes the methylation modification at K4 lysine on H3 polypeptide substrate, making it a substrate without methylation...More data for this Ligand-Target Pair
Affinity DataIC50: 25nMAssay Description:Principle: LSD1 specifically removes the methylation modification at K4 lysine on H3 polypeptide substrate, making it a substrate without methylation...More data for this Ligand-Target Pair
Affinity DataIC50: 25.6nMAssay Description:Principle: LSD1 specifically removes the methylation modification at K4 lysine on H3 polypeptide substrate, making it a substrate without methylation...More data for this Ligand-Target Pair
Affinity DataIC50: 26.9nMAssay Description:Principle: LSD1 specifically removes the methylation modification at K4 lysine on H3 polypeptide substrate, making it a substrate without methylation...More data for this Ligand-Target Pair
Affinity DataIC50: 28.2nMAssay Description:Principle: LSD1 specifically removes the methylation modification at K4 lysine on H3 polypeptide substrate, making it a substrate without methylation...More data for this Ligand-Target Pair
Affinity DataIC50: 28.8nMAssay Description:Principle: LSD1 specifically removes the methylation modification at K4 lysine on H3 polypeptide substrate, making it a substrate without methylation...More data for this Ligand-Target Pair
Affinity DataIC50: 29.9nMAssay Description:Principle: LSD1 specifically removes the methylation modification at K4 lysine on H3 polypeptide substrate, making it a substrate without methylation...More data for this Ligand-Target Pair
Affinity DataIC50: 33.6nMAssay Description:Principle: LSD1 specifically removes the methylation modification at K4 lysine on H3 polypeptide substrate, making it a substrate without methylation...More data for this Ligand-Target Pair
Affinity DataIC50: 33.6nMAssay Description:Principle: LSD1 specifically removes the methylation modification at K4 lysine on H3 polypeptide substrate, making it a substrate without methylation...More data for this Ligand-Target Pair
Affinity DataIC50: 35.3nMAssay Description:Principle: LSD1 specifically removes the methylation modification at K4 lysine on H3 polypeptide substrate, making it a substrate without methylation...More data for this Ligand-Target Pair
Affinity DataIC50: 38.2nMAssay Description:Principle: LSD1 specifically removes the methylation modification at K4 lysine on H3 polypeptide substrate, making it a substrate without methylation...More data for this Ligand-Target Pair
Affinity DataIC50: 38.9nMAssay Description:Principle: LSD1 specifically removes the methylation modification at K4 lysine on H3 polypeptide substrate, making it a substrate without methylation...More data for this Ligand-Target Pair
Affinity DataIC50: 39.5nMAssay Description:Principle: LSD1 specifically removes the methylation modification at K4 lysine on H3 polypeptide substrate, making it a substrate without methylation...More data for this Ligand-Target Pair
Affinity DataIC50: 41.6nMAssay Description:Principle: LSD1 specifically removes the methylation modification at K4 lysine on H3 polypeptide substrate, making it a substrate without methylation...More data for this Ligand-Target Pair
Affinity DataIC50: 42.5nMAssay Description:Principle: LSD1 specifically removes the methylation modification at K4 lysine on H3 polypeptide substrate, making it a substrate without methylation...More data for this Ligand-Target Pair
Affinity DataIC50: 43.3nMAssay Description:Principle: LSD1 specifically removes the methylation modification at K4 lysine on H3 polypeptide substrate, making it a substrate without methylation...More data for this Ligand-Target Pair
Affinity DataIC50: 43.6nMAssay Description:Principle: LSD1 specifically removes the methylation modification at K4 lysine on H3 polypeptide substrate, making it a substrate without methylation...More data for this Ligand-Target Pair
Affinity DataIC50: 47.5nMAssay Description:Principle: LSD1 specifically removes the methylation modification at K4 lysine on H3 polypeptide substrate, making it a substrate without methylation...More data for this Ligand-Target Pair
Affinity DataIC50: 48.6nMAssay Description:Principle: LSD1 specifically removes the methylation modification at K4 lysine on H3 polypeptide substrate, making it a substrate without methylation...More data for this Ligand-Target Pair
Affinity DataIC50: 50.3nMAssay Description:Principle: LSD1 specifically removes the methylation modification at K4 lysine on H3 polypeptide substrate, making it a substrate without methylation...More data for this Ligand-Target Pair
Affinity DataIC50: 62.2nMAssay Description:Principle: LSD1 specifically removes the methylation modification at K4 lysine on H3 polypeptide substrate, making it a substrate without methylation...More data for this Ligand-Target Pair
Affinity DataIC50: 62.9nMAssay Description:Principle: LSD1 specifically removes the methylation modification at K4 lysine on H3 polypeptide substrate, making it a substrate without methylation...More data for this Ligand-Target Pair
Affinity DataIC50: 68.2nMAssay Description:Principle: LSD1 specifically removes the methylation modification at K4 lysine on H3 polypeptide substrate, making it a substrate without methylation...More data for this Ligand-Target Pair
Affinity DataIC50: 71.3nMAssay Description:Principle: LSD1 specifically removes the methylation modification at K4 lysine on H3 polypeptide substrate, making it a substrate without methylation...More data for this Ligand-Target Pair
Affinity DataIC50: 85.1nMAssay Description:Principle: LSD1 specifically removes the methylation modification at K4 lysine on H3 polypeptide substrate, making it a substrate without methylation...More data for this Ligand-Target Pair
Affinity DataIC50: 164nMAssay Description:Principle: LSD1 specifically removes the methylation modification at K4 lysine on H3 polypeptide substrate, making it a substrate without methylation...More data for this Ligand-Target Pair
Affinity DataIC50: 223nMAssay Description:Principle: LSD1 specifically removes the methylation modification at K4 lysine on H3 polypeptide substrate, making it a substrate without methylation...More data for this Ligand-Target Pair
Affinity DataIC50: 261nMAssay Description:Principle: LSD1 specifically removes the methylation modification at K4 lysine on H3 polypeptide substrate, making it a substrate without methylation...More data for this Ligand-Target Pair
Affinity DataIC50: 265nMAssay Description:Principle: LSD1 specifically removes the methylation modification at K4 lysine on H3 polypeptide substrate, making it a substrate without methylation...More data for this Ligand-Target Pair
Affinity DataIC50: 334nMAssay Description:Principle: LSD1 specifically removes the methylation modification at K4 lysine on H3 polypeptide substrate, making it a substrate without methylation...More data for this Ligand-Target Pair
Affinity DataIC50: 354nMAssay Description:Principle: LSD1 specifically removes the methylation modification at K4 lysine on H3 polypeptide substrate, making it a substrate without methylation...More data for this Ligand-Target Pair
Affinity DataIC50: 404nMAssay Description:Principle: LSD1 specifically removes the methylation modification at K4 lysine on H3 polypeptide substrate, making it a substrate without methylation...More data for this Ligand-Target Pair
Affinity DataIC50: 461nMAssay Description:Principle: LSD1 specifically removes the methylation modification at K4 lysine on H3 polypeptide substrate, making it a substrate without methylation...More data for this Ligand-Target Pair
Affinity DataIC50: 534nMAssay Description:Principle: LSD1 specifically removes the methylation modification at K4 lysine on H3 polypeptide substrate, making it a substrate without methylation...More data for this Ligand-Target Pair
Affinity DataIC50: 1.69E+3nMAssay Description:Principle: LSD1 specifically removes the methylation modification at K4 lysine on H3 polypeptide substrate, making it a substrate without methylation...More data for this Ligand-Target Pair
Affinity DataIC50: 1.79E+3nMAssay Description:Principle: LSD1 specifically removes the methylation modification at K4 lysine on H3 polypeptide substrate, making it a substrate without methylation...More data for this Ligand-Target Pair
Affinity DataIC50: 3.14E+3nMAssay Description:Principle: A specific luciferin derivative was used as a substrate. MAOA or MAOB can catalyze the conversion of substrate to luciferin methyl ester. ...More data for this Ligand-Target Pair
Affinity DataIC50: 7.33E+3nMAssay Description:Principle: A specific luciferin derivative was used as a substrate. MAOA or MAOB can catalyze the conversion of substrate to luciferin methyl ester. ...More data for this Ligand-Target Pair
Affinity DataIC50: 9.22E+3nMAssay Description:Principle: A specific luciferin derivative was used as a substrate. MAOA or MAOB can catalyze the conversion of substrate to luciferin methyl ester. ...More data for this Ligand-Target Pair
Affinity DataIC50: 1.07E+4nMAssay Description:Principle: A specific luciferin derivative was used as a substrate. MAOA or MAOB can catalyze the conversion of substrate to luciferin methyl ester. ...More data for this Ligand-Target Pair
Affinity DataIC50: 1.59E+4nMAssay Description:Principle: A specific luciferin derivative was used as a substrate. MAOA or MAOB can catalyze the conversion of substrate to luciferin methyl ester. ...More data for this Ligand-Target Pair
Affinity DataIC50: 1.62E+4nMAssay Description:Principle: A specific luciferin derivative was used as a substrate. MAOA or MAOB can catalyze the conversion of substrate to luciferin methyl ester. ...More data for this Ligand-Target Pair
Affinity DataIC50: 3.29E+4nMAssay Description:Principle: A specific luciferin derivative was used as a substrate. MAOA or MAOB can catalyze the conversion of substrate to luciferin methyl ester. ...More data for this Ligand-Target Pair
Affinity DataIC50: 3.55E+4nMAssay Description:Principle: A specific luciferin derivative was used as a substrate. MAOA or MAOB can catalyze the conversion of substrate to luciferin methyl ester. ...More data for this Ligand-Target Pair