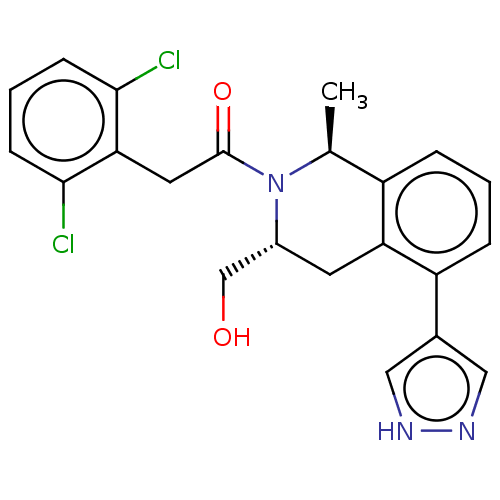
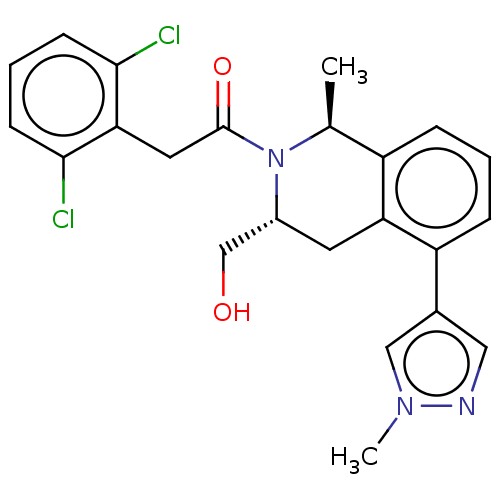
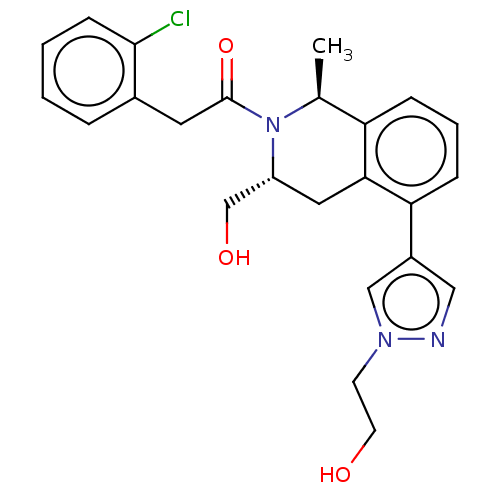
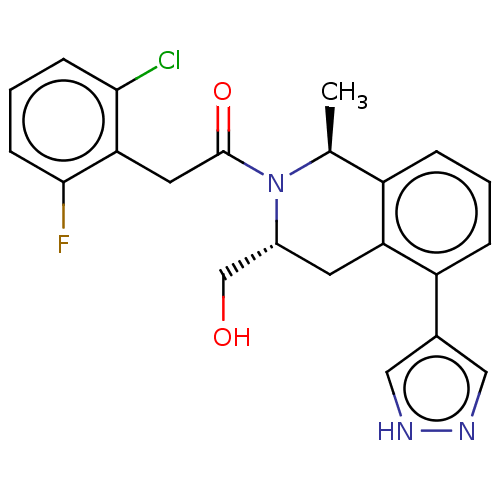
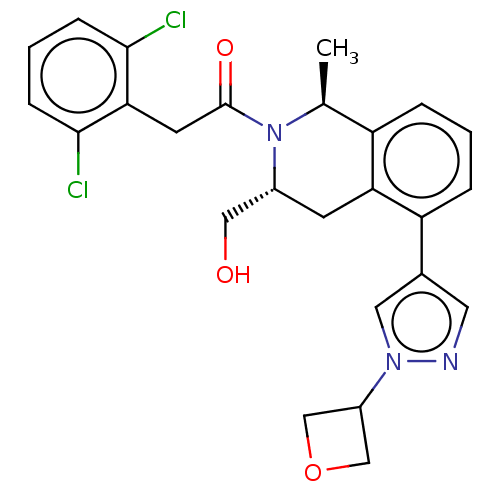
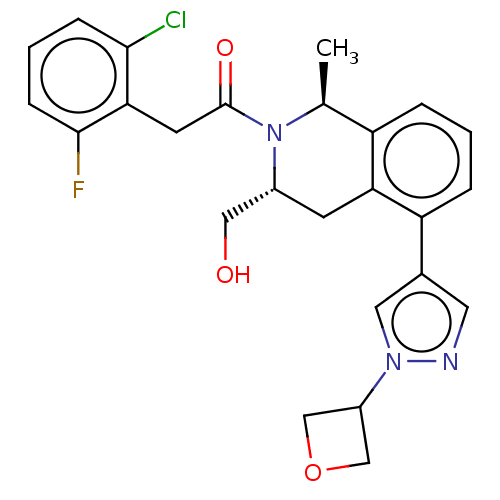
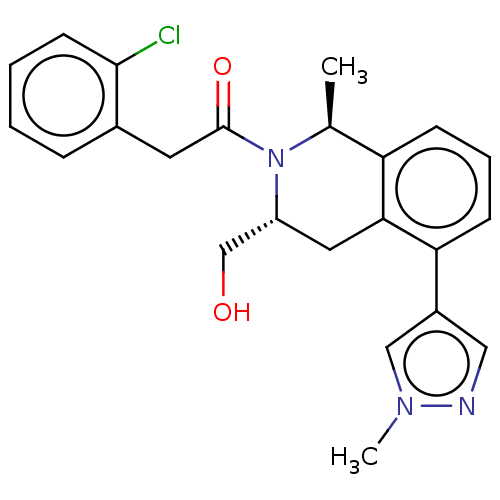
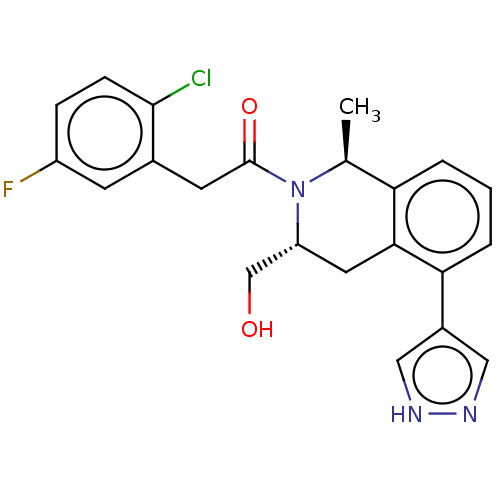
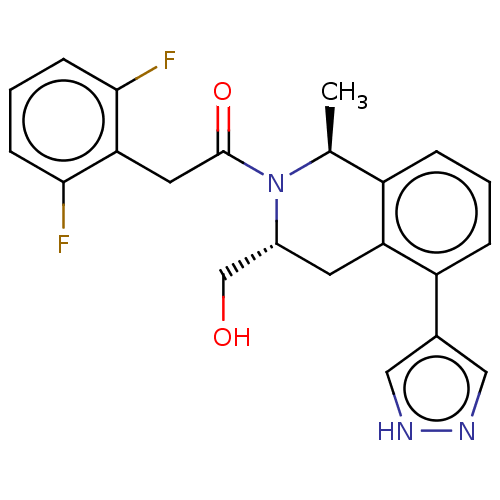
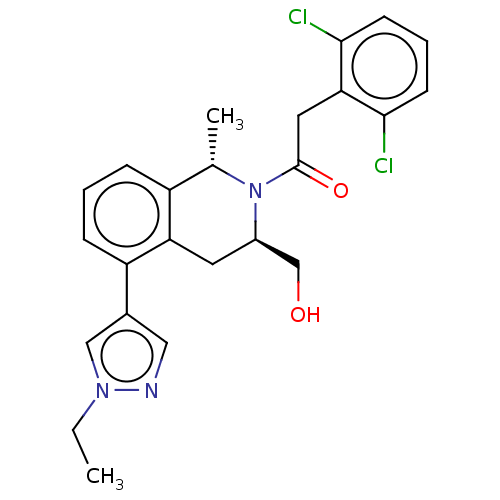
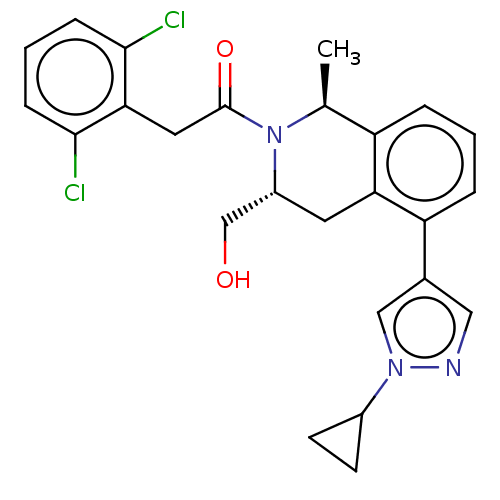
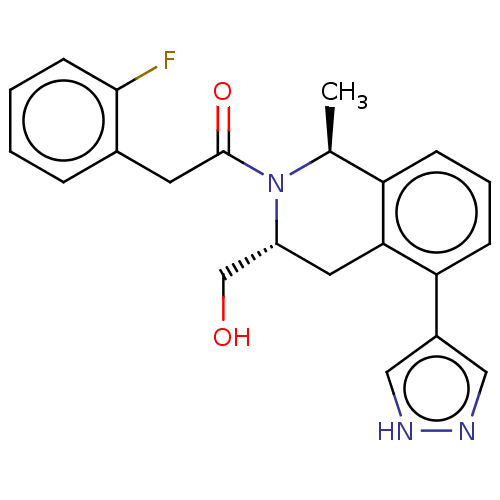
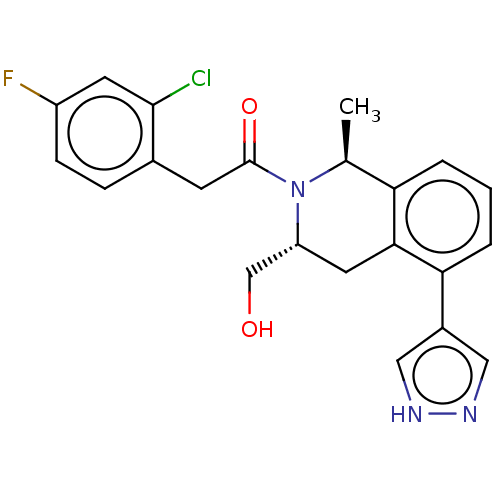
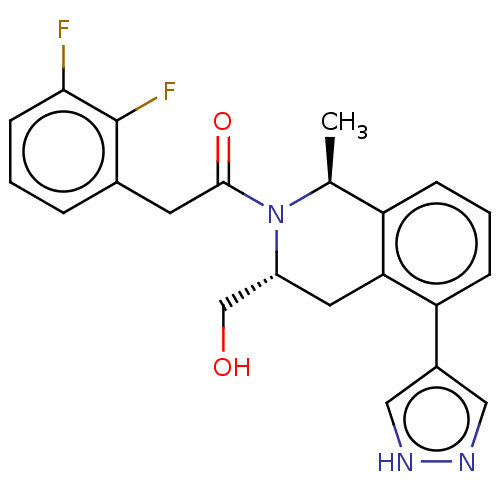
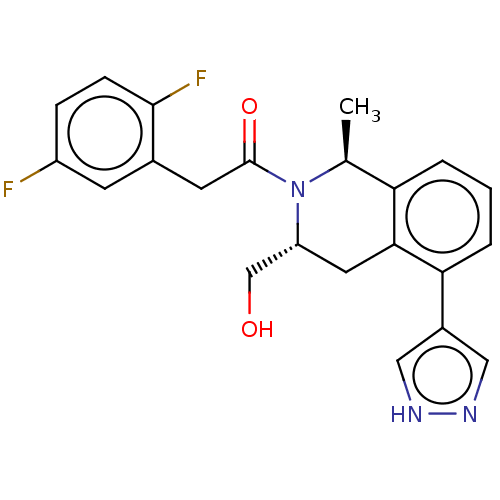
Report error Found 15 Enz. Inhib. hit(s) with all data for entry = 9025
Affinity DataEC50: 13.4nMAssay Description:The PAM activity of the compounds of the present invention may be measured essentially as described in Svensson et al., An Allosteric Potentiator of ...More data for this Ligand-Target Pair
Affinity DataEC50: 16.4nMAssay Description:The PAM activity of the compounds of the present invention may be measured essentially as described in Svensson et al., An Allosteric Potentiator of ...More data for this Ligand-Target Pair
Affinity DataEC50: 18.3nMAssay Description:The PAM activity of the compounds of the present invention may be measured essentially as described in Svensson et al., An Allosteric Potentiator of ...More data for this Ligand-Target Pair
Affinity DataEC50: 28nMAssay Description:The PAM activity of the compounds of the present invention may be measured essentially as described in Svensson et al., An Allosteric Potentiator of ...More data for this Ligand-Target Pair
Affinity DataEC50: 28.5nMAssay Description:The PAM activity of the compounds of the present invention may be measured essentially as described in Svensson et al., An Allosteric Potentiator of ...More data for this Ligand-Target Pair
Affinity DataEC50: 42.2nMAssay Description:The PAM activity of the compounds of the present invention may be measured essentially as described in Svensson et al., An Allosteric Potentiator of ...More data for this Ligand-Target Pair
Affinity DataEC50: 81.2nMAssay Description:The PAM activity of the compounds of the present invention may be measured essentially as described in Svensson et al., An Allosteric Potentiator of ...More data for this Ligand-Target Pair
Affinity DataEC50: 112nMAssay Description:The PAM activity of the compounds of the present invention may be measured essentially as described in Svensson et al., An Allosteric Potentiator of ...More data for this Ligand-Target Pair
Affinity DataEC50: 127nMAssay Description:The PAM activity of the compounds of the present invention may be measured essentially as described in Svensson et al., An Allosteric Potentiator of ...More data for this Ligand-Target Pair
Affinity DataEC50: 172nMAssay Description:The PAM activity of the compounds of the present invention may be measured essentially as described in Svensson et al., An Allosteric Potentiator of ...More data for this Ligand-Target Pair
Affinity DataEC50: 287nMAssay Description:The PAM activity of the compounds of the present invention may be measured essentially as described in Svensson et al., An Allosteric Potentiator of ...More data for this Ligand-Target Pair
Affinity DataEC50: 484nMAssay Description:The PAM activity of the compounds of the present invention may be measured essentially as described in Svensson et al., An Allosteric Potentiator of ...More data for this Ligand-Target Pair
Affinity DataEC50: 540nMAssay Description:The PAM activity of the compounds of the present invention may be measured essentially as described in Svensson et al., An Allosteric Potentiator of ...More data for this Ligand-Target Pair
Affinity DataEC50: 677nMAssay Description:The PAM activity of the compounds of the present invention may be measured essentially as described in Svensson et al., An Allosteric Potentiator of ...More data for this Ligand-Target Pair
Affinity DataEC50: 836nMAssay Description:The PAM activity of the compounds of the present invention may be measured essentially as described in Svensson et al., An Allosteric Potentiator of ...More data for this Ligand-Target Pair