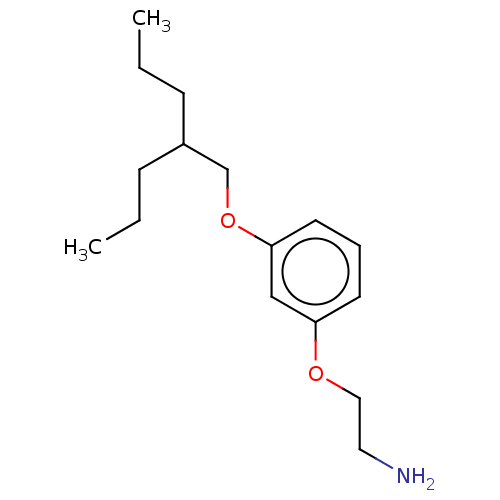
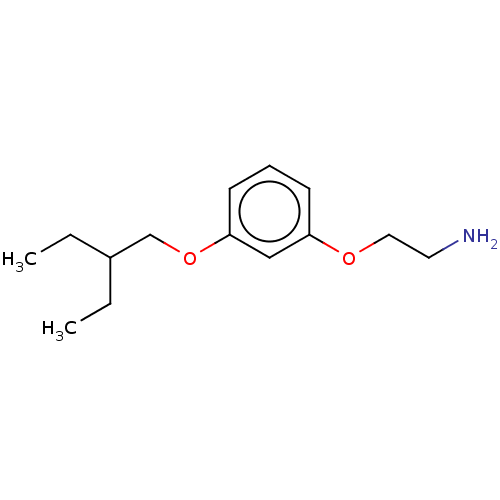
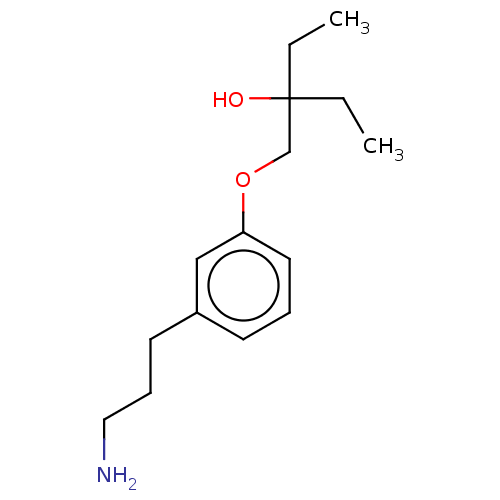
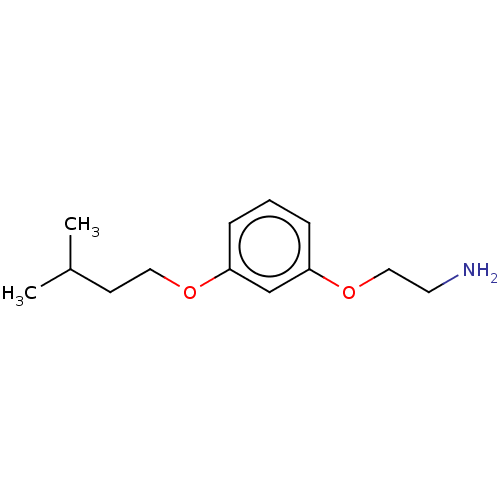
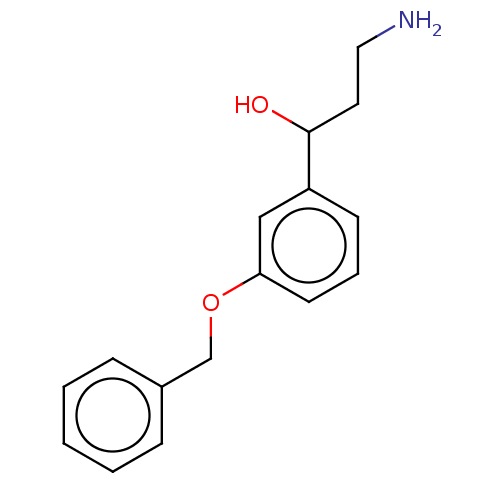
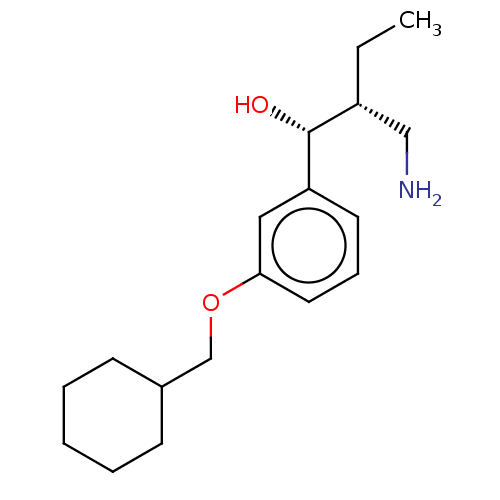
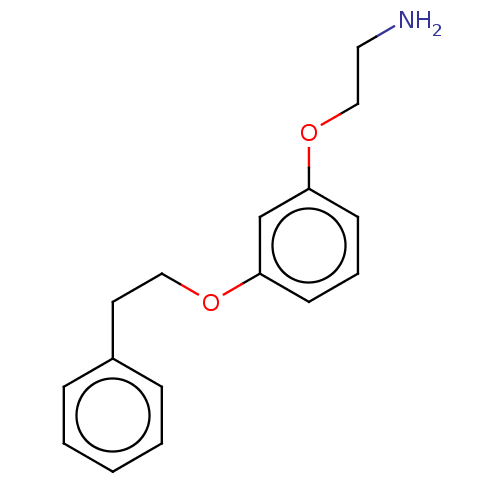
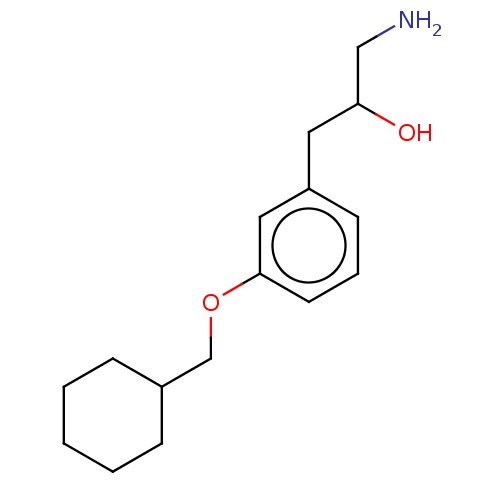
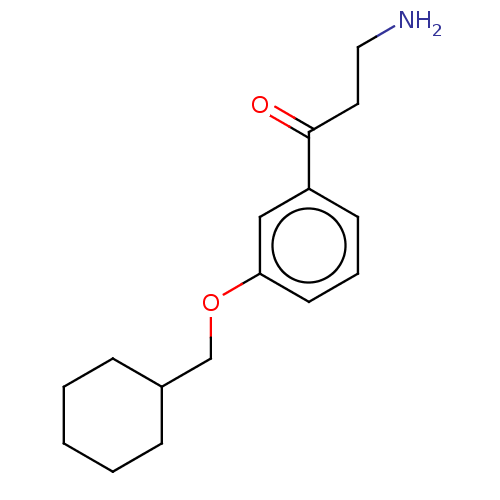
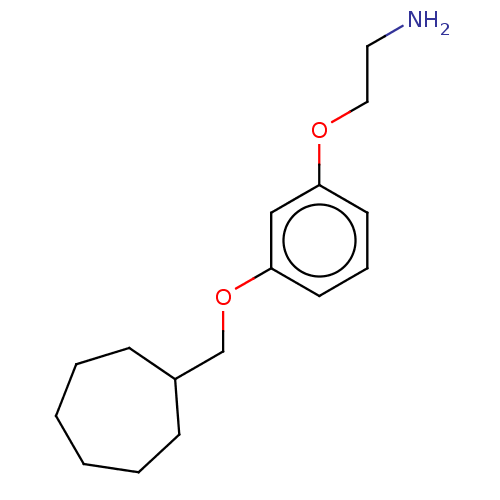
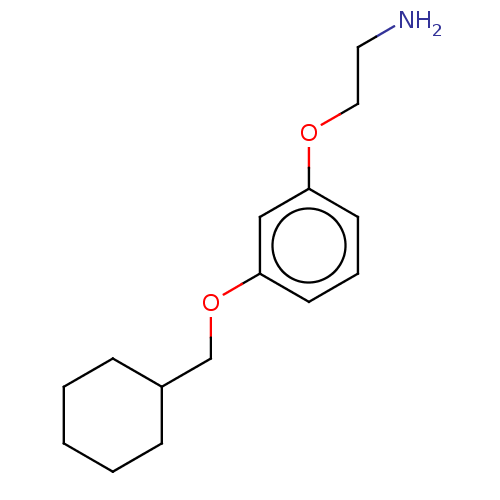
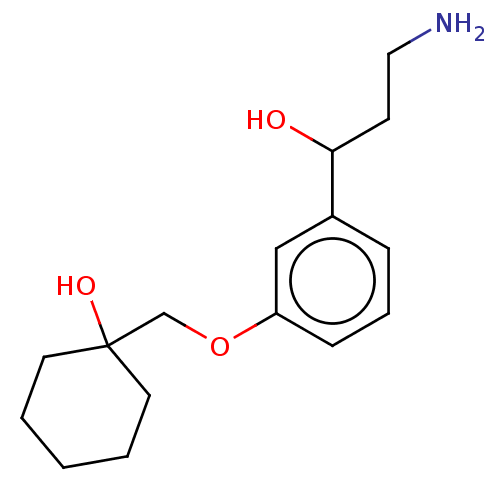
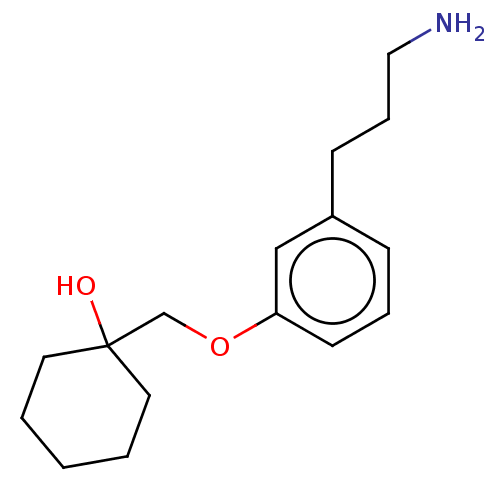
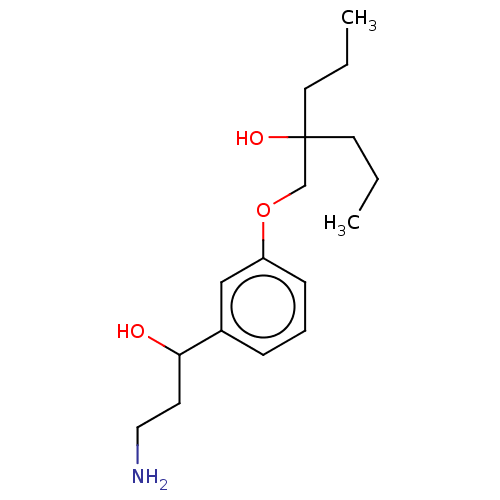
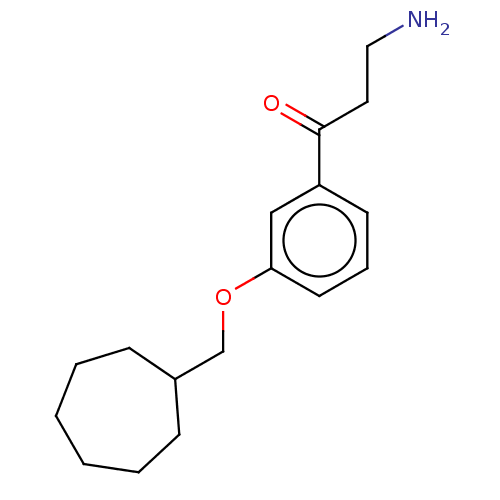
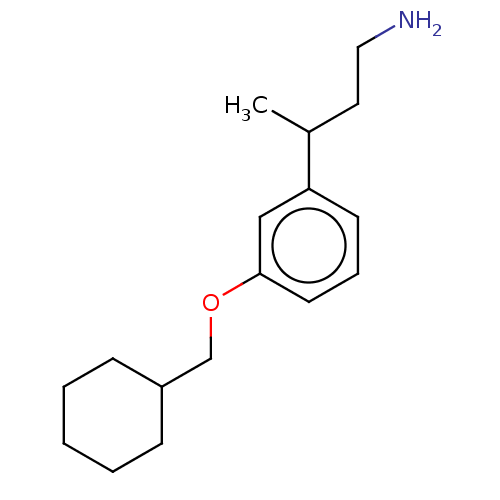
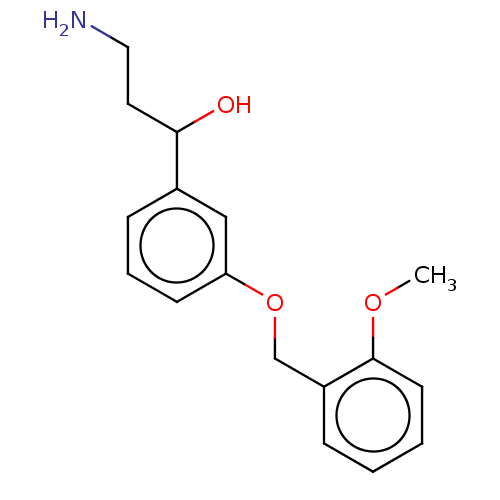
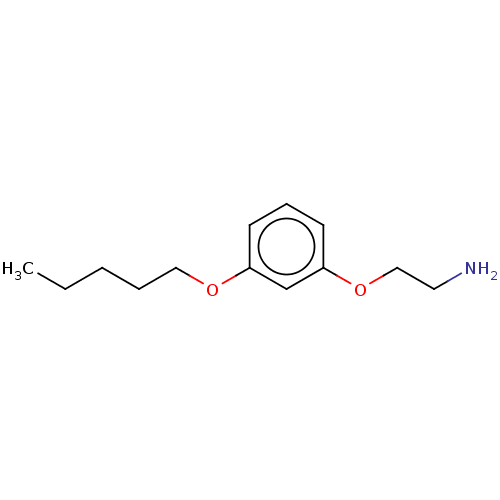
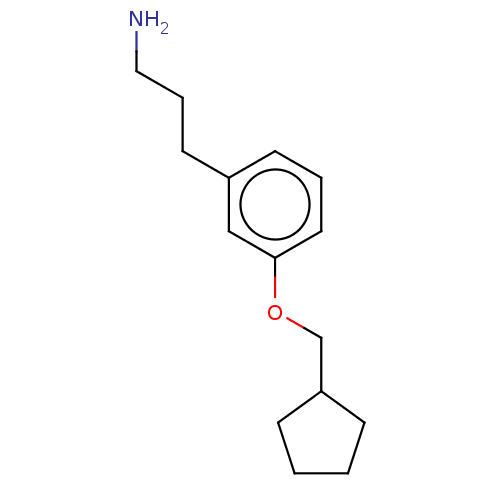
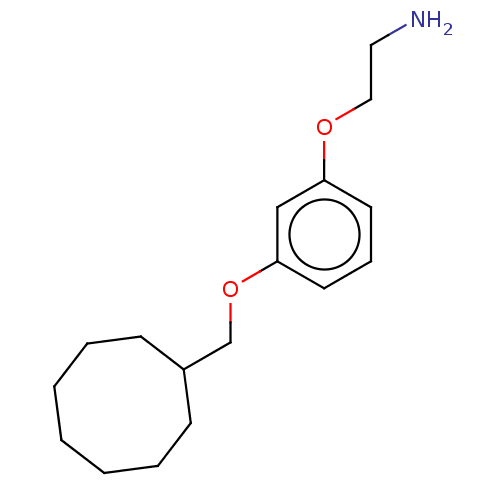
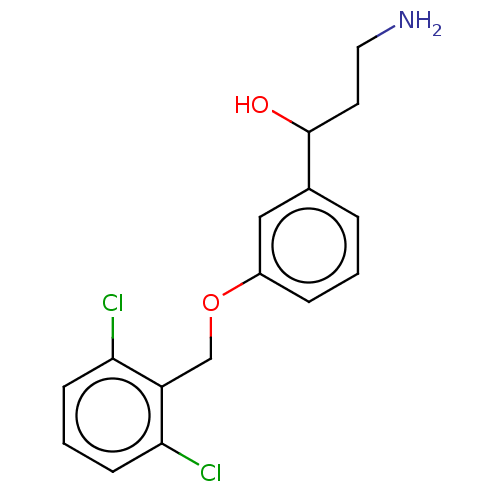
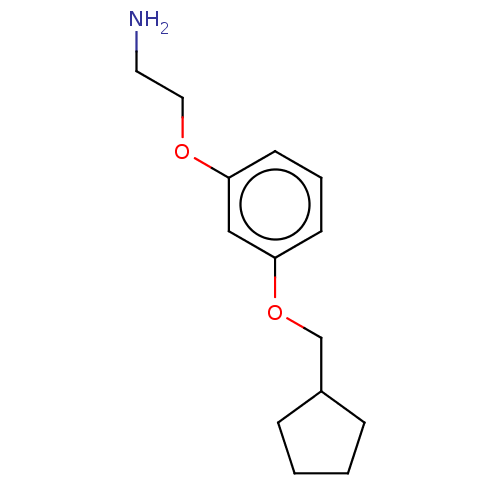
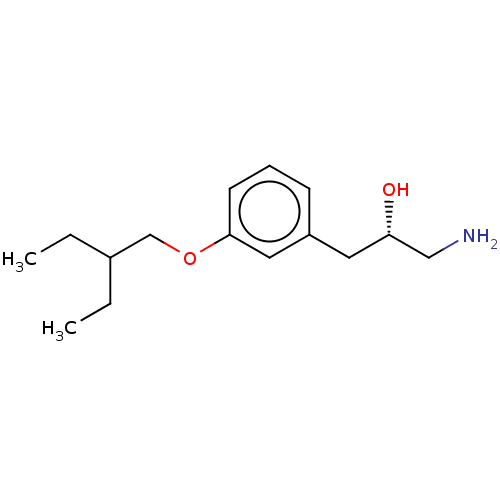
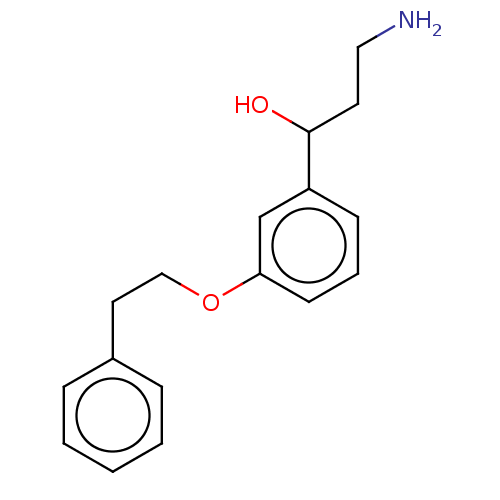
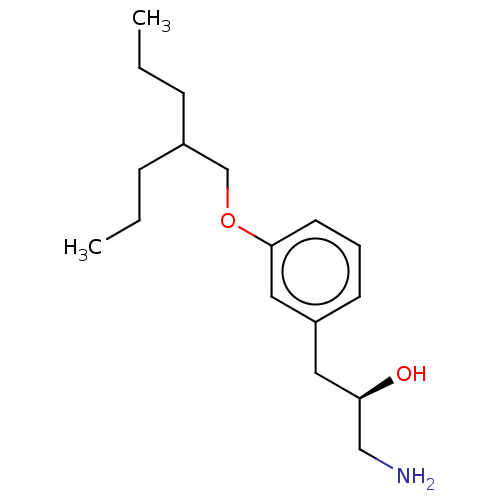
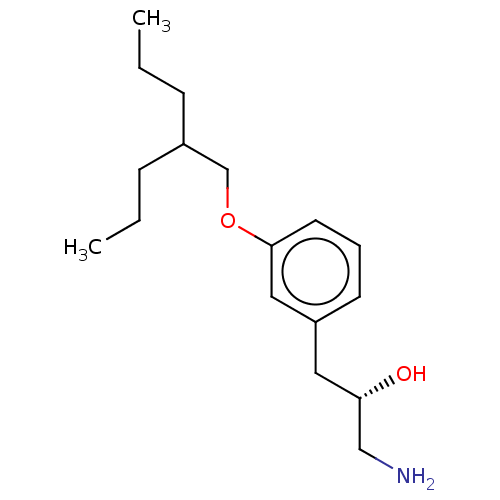
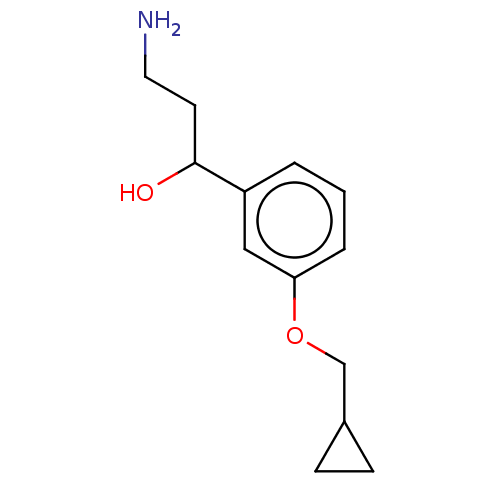
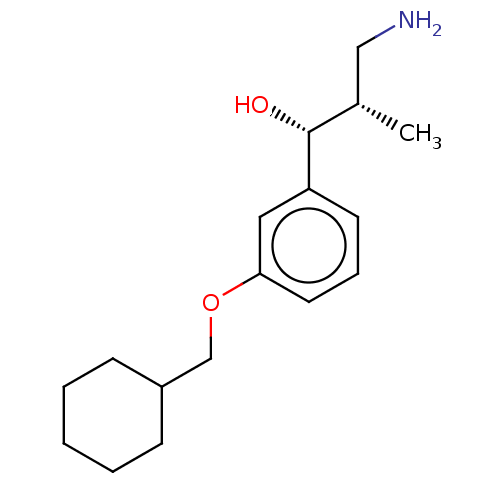
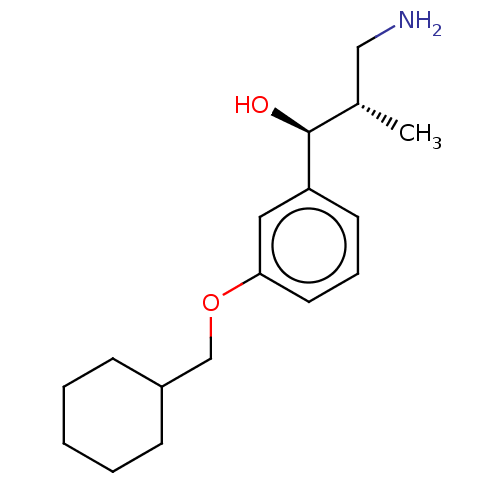
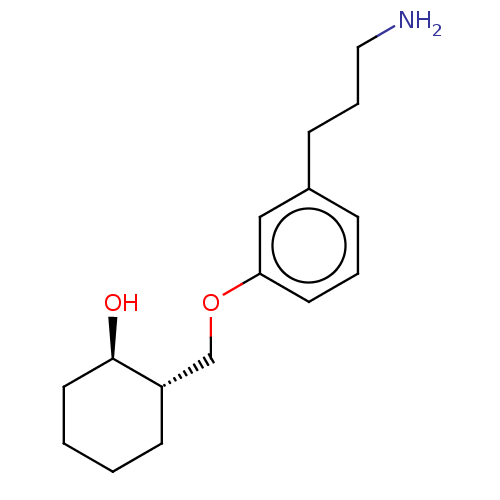
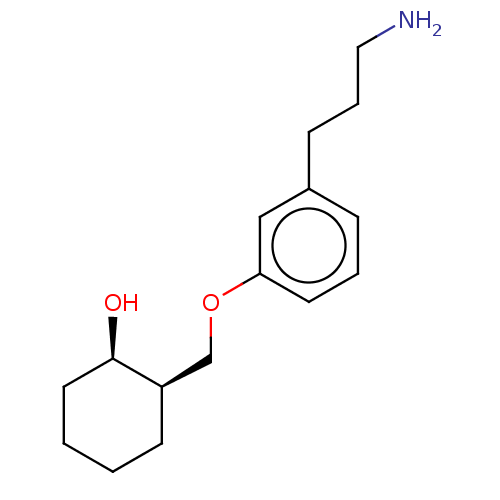
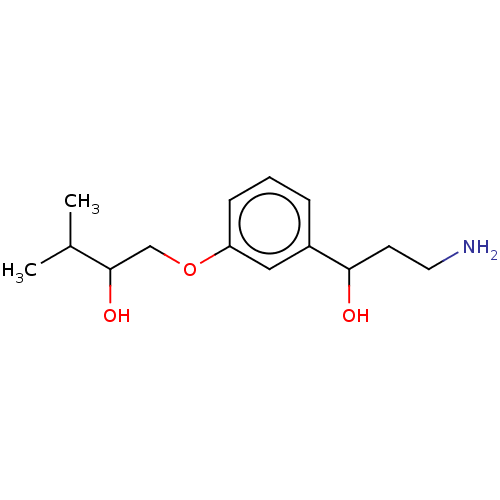
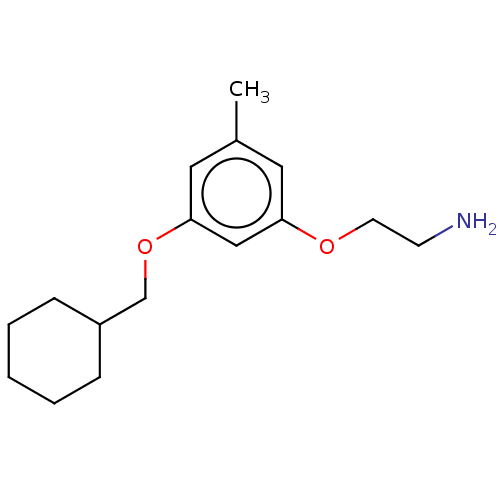
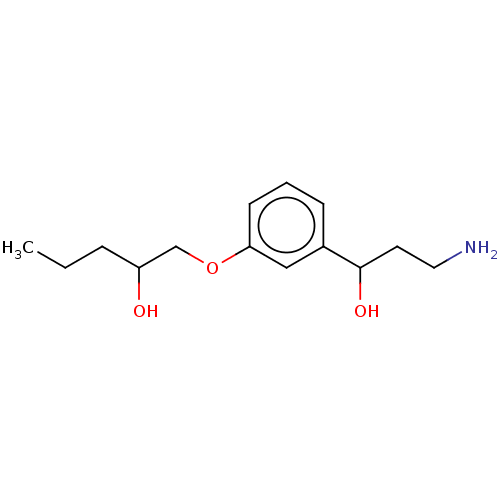
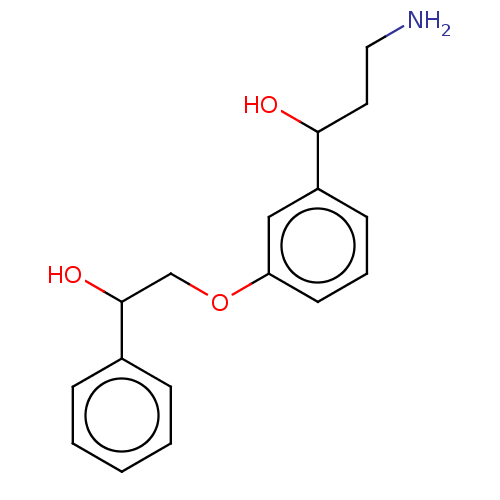
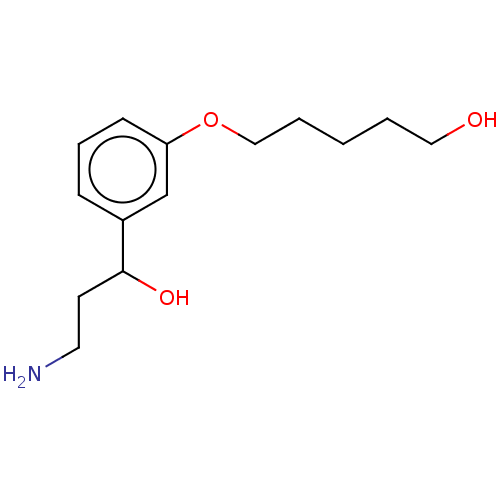
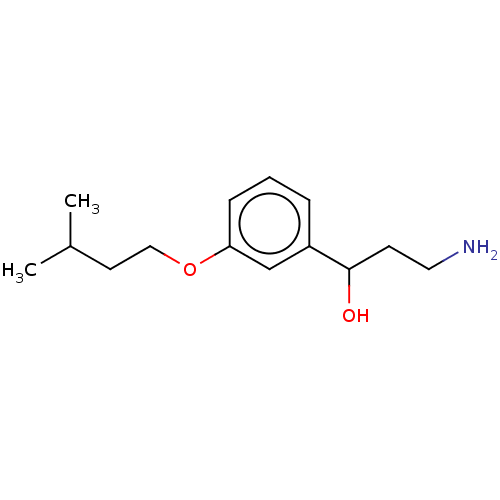
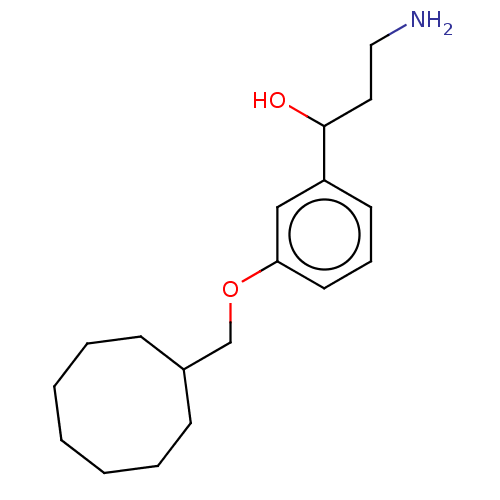
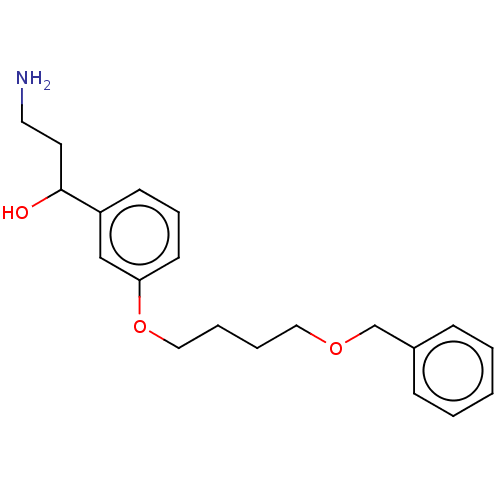
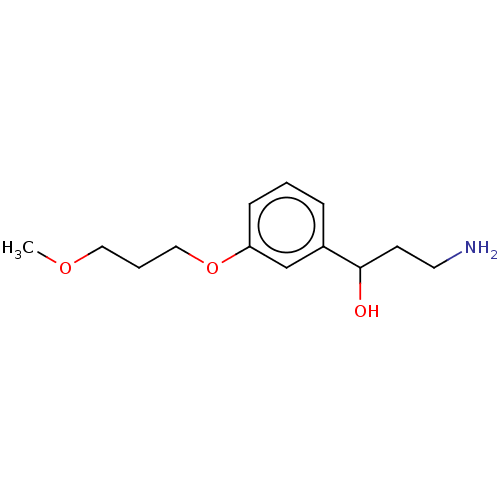
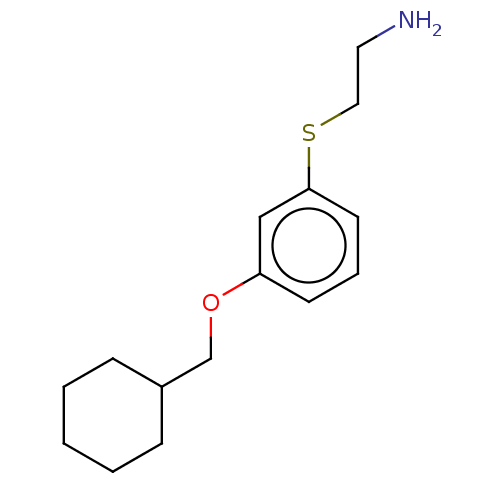
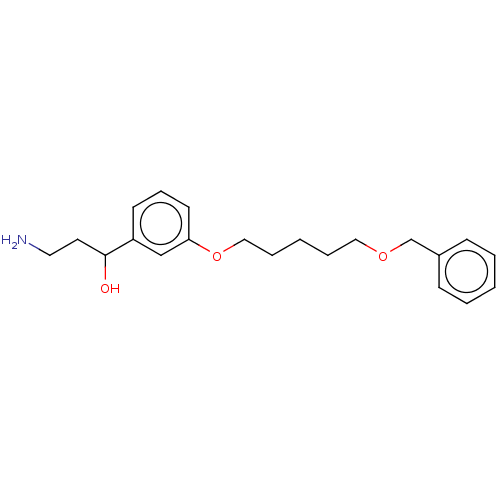
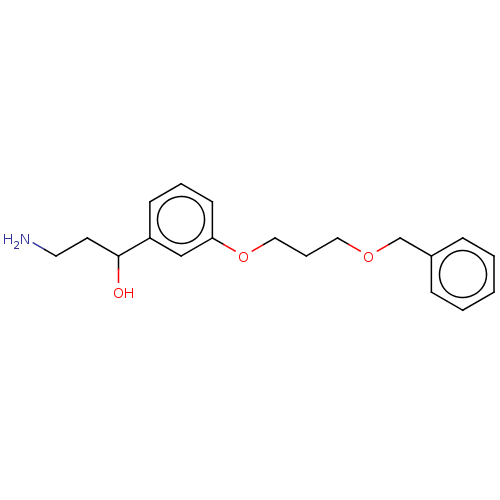
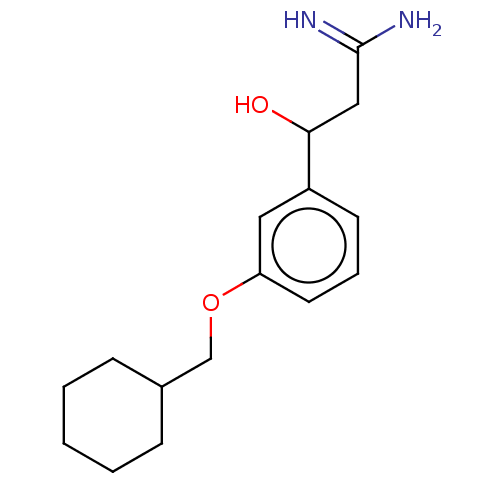
Report error Found 195 Enz. Inhib. hit(s) with all data for entry = 1446
Affinity DataEC50: 0.0650nMAssay Description:Retinoid nuclear receptor activity is associated with transduction of the non-visual physiologic, pharmacologic, and toxicologic retinoid signals tha...More data for this Ligand-Target Pair
Affinity DataEC50: 0.300nMAssay Description:Retinoid nuclear receptor activity is associated with transduction of the non-visual physiologic, pharmacologic, and toxicologic retinoid signals tha...More data for this Ligand-Target Pair
Affinity DataEC50: 5.5nMAssay Description:Retinoid nuclear receptor activity is associated with transduction of the non-visual physiologic, pharmacologic, and toxicologic retinoid signals tha...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:Isomerase inhibition reactions were performed essentially as described (Stecher et al., J. Biol. Chem. 274:8577-85 (1999); see also Golczak et al., P...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:Isomerase inhibition reactions were performed essentially as described (Stecher et al., J. Biol. Chem. 274:8577-85 (1999); see also Golczak et al., P...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:Isomerase inhibition reactions were performed essentially as described (Stecher et al., J. Biol. Chem. 274:8577-85 (1999); see also Golczak et al., P...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:Isomerase inhibition reactions were performed essentially as described (Stecher et al., J. Biol. Chem. 274:8577-85 (1999); see also Golczak et al., P...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:Isomerase inhibition reactions were performed essentially as described (Stecher et al., J. Biol. Chem. 274:8577-85 (1999); see also Golczak et al., P...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:Isomerase inhibition reactions were performed essentially as described (Stecher et al., J. Biol. Chem. 274:8577-85 (1999); see also Golczak et al., P...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:Isomerase inhibition reactions were performed essentially as described (Stecher et al., J. Biol. Chem. 274:8577-85 (1999); see also Golczak et al., P...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:Isomerase inhibition reactions were performed essentially as described (Stecher et al., J. Biol. Chem. 274:8577-85 (1999); see also Golczak et al., P...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:Isomerase inhibition reactions were performed essentially as described (Stecher et al., J. Biol. Chem. 274:8577-85 (1999); see also Golczak et al., P...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:Isomerase inhibition reactions were performed essentially as described (Stecher et al., J. Biol. Chem. 274:8577-85 (1999); see also Golczak et al., P...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:Isomerase inhibition reactions were performed essentially as described (Stecher et al., J. Biol. Chem. 274:8577-85 (1999); see also Golczak et al., P...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:Isomerase inhibition reactions were performed essentially as described (Stecher et al., J. Biol. Chem. 274:8577-85 (1999); see also Golczak et al., P...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:Isomerase inhibition reactions were performed essentially as described (Stecher et al., J. Biol. Chem. 274:8577-85 (1999); see also Golczak et al., P...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:Isomerase inhibition reactions were performed essentially as described (Stecher et al., J. Biol. Chem. 274:8577-85 (1999); see also Golczak et al., P...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:Isomerase inhibition reactions were performed essentially as described (Stecher et al., J. Biol. Chem. 274:8577-85 (1999); see also Golczak et al., P...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:Isomerase inhibition reactions were performed essentially as described (Stecher et al., J. Biol. Chem. 274:8577-85 (1999); see also Golczak et al., P...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:Isomerase inhibition reactions were performed essentially as described (Stecher et al., J. Biol. Chem. 274:8577-85 (1999); see also Golczak et al., P...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:Isomerase inhibition reactions were performed essentially as described (Stecher et al., J. Biol. Chem. 274:8577-85 (1999); see also Golczak et al., P...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:Isomerase inhibition reactions were performed essentially as described (Stecher et al., J. Biol. Chem. 274:8577-85 (1999); see also Golczak et al., P...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:Isomerase inhibition reactions were performed essentially as described (Stecher et al., J. Biol. Chem. 274:8577-85 (1999); see also Golczak et al., P...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:Isomerase inhibition reactions were performed essentially as described (Stecher et al., J. Biol. Chem. 274:8577-85 (1999); see also Golczak et al., P...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:Isomerase inhibition reactions were performed essentially as described (Stecher et al., J. Biol. Chem. 274:8577-85 (1999); see also Golczak et al., P...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:Isomerase inhibition reactions were performed essentially as described (Stecher et al., J. Biol. Chem. 274:8577-85 (1999); see also Golczak et al., P...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:Isomerase inhibition reactions were performed essentially as described (Stecher et al., J. Biol. Chem. 274:8577-85 (1999); see also Golczak et al., P...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:Isomerase inhibition reactions were performed essentially as described (Stecher et al., J. Biol. Chem. 274:8577-85 (1999); see also Golczak et al., P...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:Isomerase inhibition reactions were performed essentially as described (Stecher et al., J. Biol. Chem. 274:8577-85 (1999); see also Golczak et al., P...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:Isomerase inhibition reactions were performed essentially as described (Stecher et al., J. Biol. Chem. 274:8577-85 (1999); see also Golczak et al., P...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:Isomerase inhibition reactions were performed essentially as described (Stecher et al., J. Biol. Chem. 274:8577-85 (1999); see also Golczak et al., P...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:Isomerase inhibition reactions were performed essentially as described (Stecher et al., J. Biol. Chem. 274:8577-85 (1999); see also Golczak et al., P...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:Isomerase inhibition reactions were performed essentially as described (Stecher et al., J. Biol. Chem. 274:8577-85 (1999); see also Golczak et al., P...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:Isomerase inhibition reactions were performed essentially as described (Stecher et al., J. Biol. Chem. 274:8577-85 (1999); see also Golczak et al., P...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:Isomerase inhibition reactions were performed essentially as described (Stecher et al., J. Biol. Chem. 274:8577-85 (1999); see also Golczak et al., P...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:Isomerase inhibition reactions were performed essentially as described (Stecher et al., J. Biol. Chem. 274:8577-85 (1999); see also Golczak et al., P...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:Isomerase inhibition reactions were performed essentially as described (Stecher et al., J. Biol. Chem. 274:8577-85 (1999); see also Golczak et al., P...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:Isomerase inhibition reactions were performed essentially as described (Stecher et al., J. Biol. Chem. 274:8577-85 (1999); see also Golczak et al., P...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:Isomerase inhibition reactions were performed essentially as described (Stecher et al., J. Biol. Chem. 274:8577-85 (1999); see also Golczak et al., P...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:Isomerase inhibition reactions were performed essentially as described (Stecher et al., J. Biol. Chem. 274:8577-85 (1999); see also Golczak et al., P...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:Isomerase inhibition reactions were performed essentially as described (Stecher et al., J. Biol. Chem. 274:8577-85 (1999); see also Golczak et al., P...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:Isomerase inhibition reactions were performed essentially as described (Stecher et al., J. Biol. Chem. 274:8577-85 (1999); see also Golczak et al., P...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:Isomerase inhibition reactions were performed essentially as described (Stecher et al., J. Biol. Chem. 274:8577-85 (1999); see also Golczak et al., P...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:Isomerase inhibition reactions were performed essentially as described (Stecher et al., J. Biol. Chem. 274:8577-85 (1999); see also Golczak et al., P...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:Isomerase inhibition reactions were performed essentially as described (Stecher et al., J. Biol. Chem. 274:8577-85 (1999); see also Golczak et al., P...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:Isomerase inhibition reactions were performed essentially as described (Stecher et al., J. Biol. Chem. 274:8577-85 (1999); see also Golczak et al., P...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:Isomerase inhibition reactions were performed essentially as described (Stecher et al., J. Biol. Chem. 274:8577-85 (1999); see also Golczak et al., P...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:Isomerase inhibition reactions were performed essentially as described (Stecher et al., J. Biol. Chem. 274:8577-85 (1999); see also Golczak et al., P...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:Isomerase inhibition reactions were performed essentially as described (Stecher et al., J. Biol. Chem. 274:8577-85 (1999); see also Golczak et al., P...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:Isomerase inhibition reactions were performed essentially as described (Stecher et al., J. Biol. Chem. 274:8577-85 (1999); see also Golczak et al., P...More data for this Ligand-Target Pair
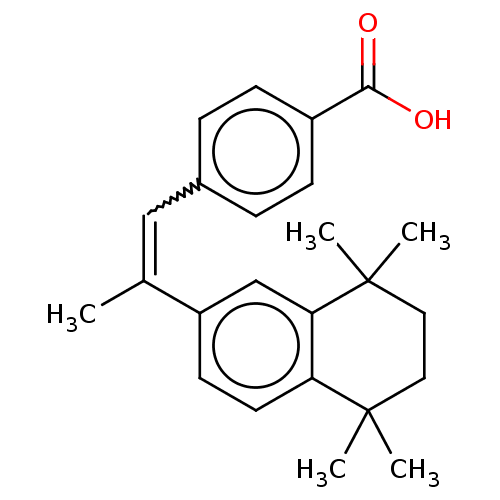
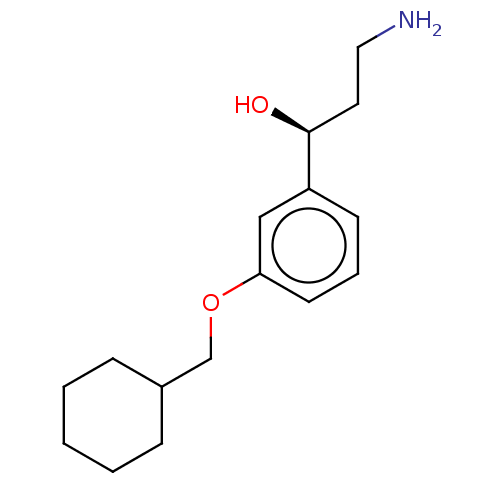
 3D Structure (crystal)
3D Structure (crystal)