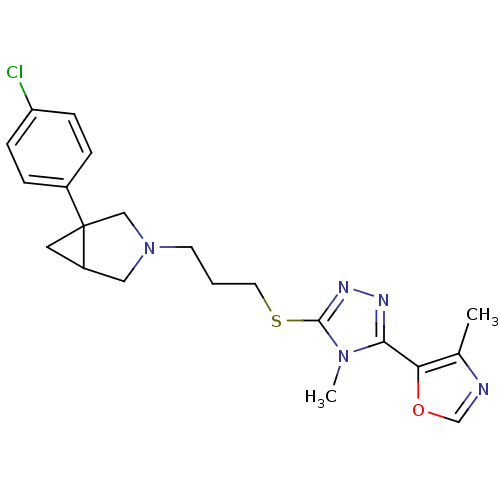
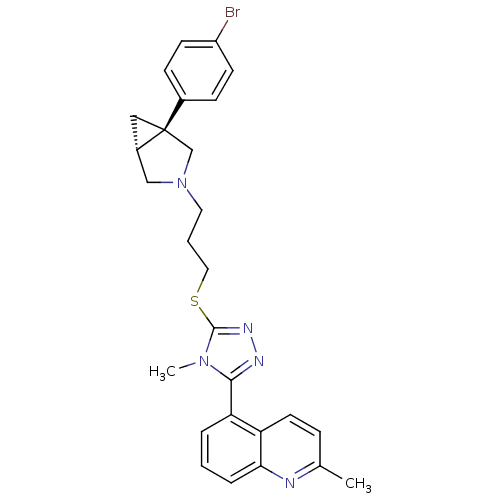
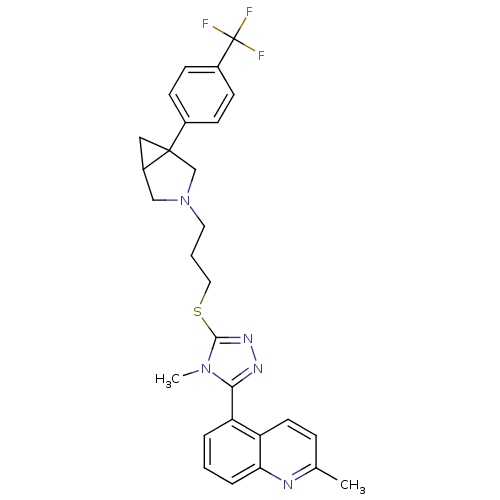
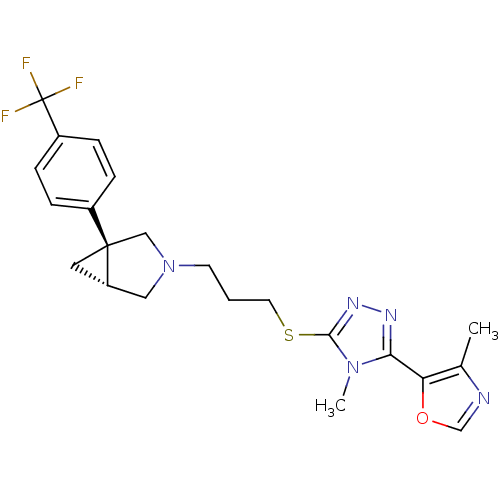
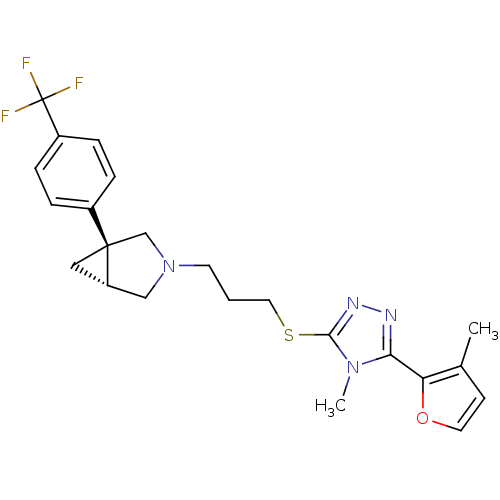
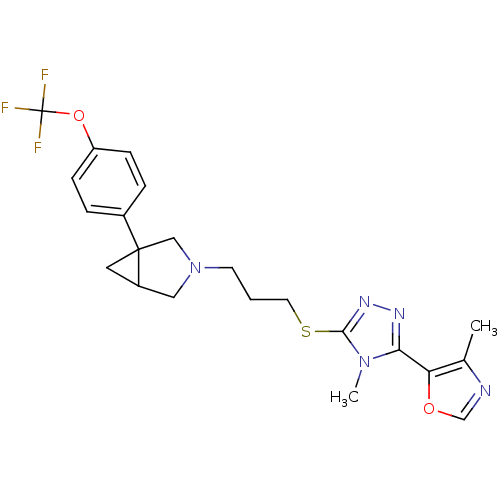
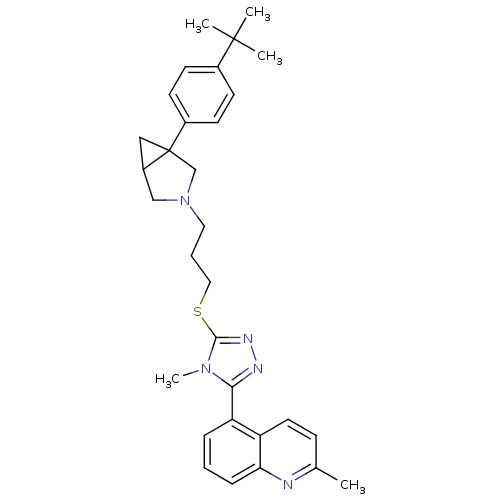
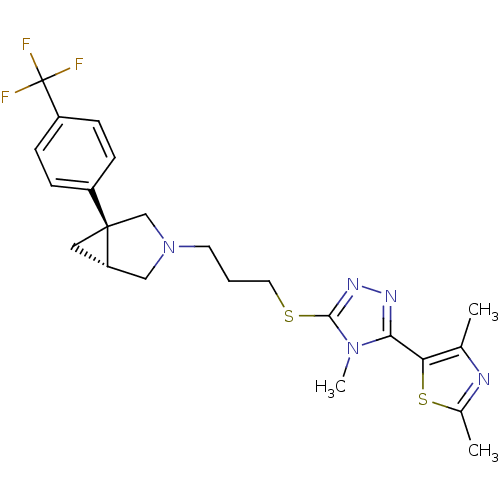
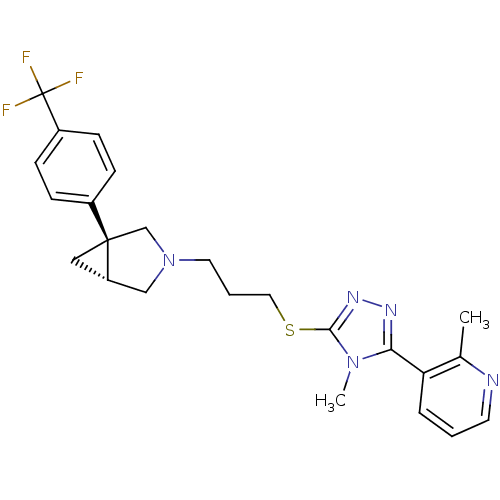
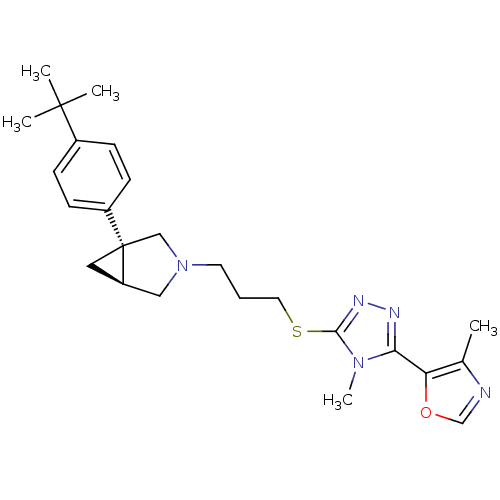
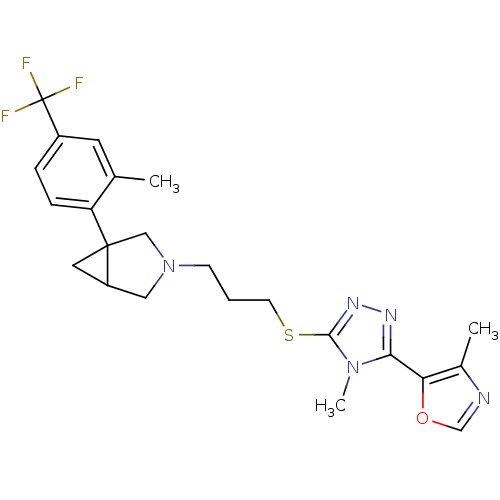
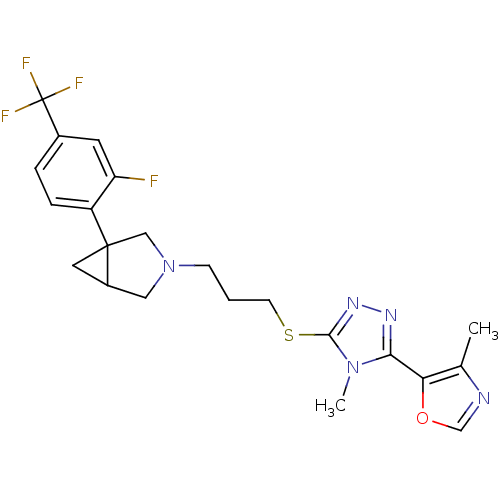
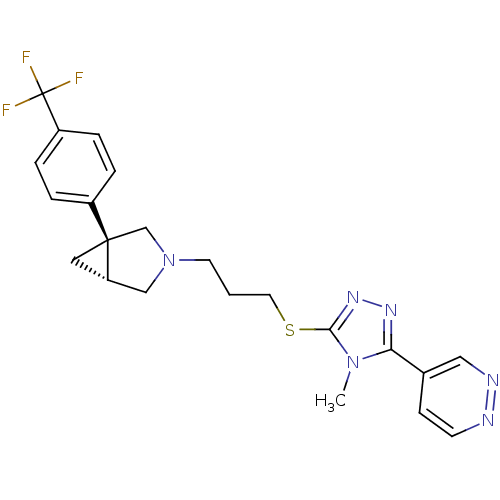
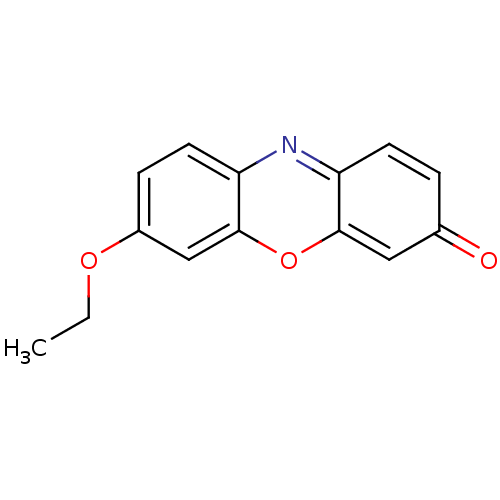
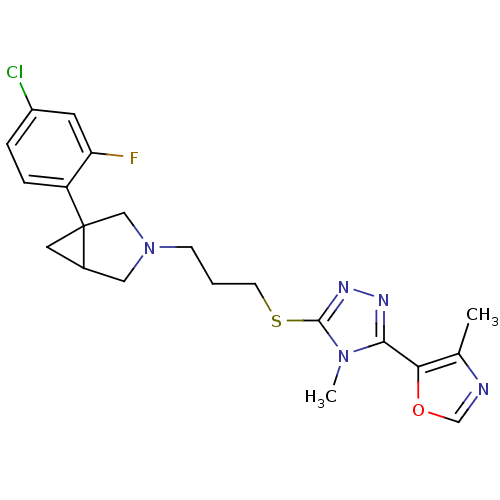
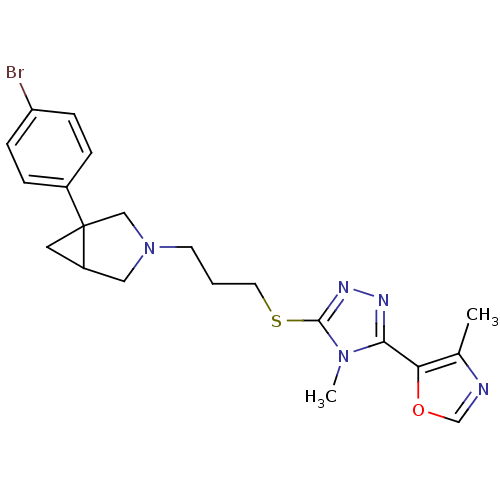
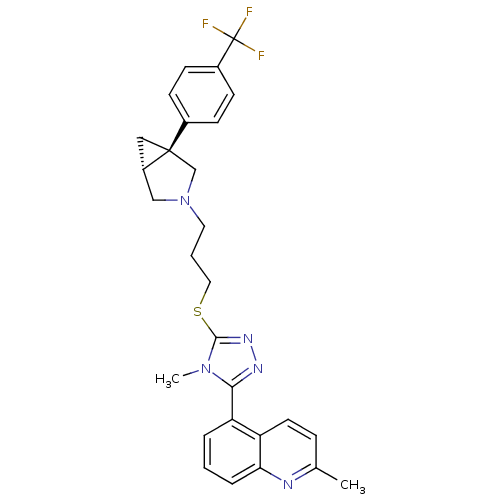
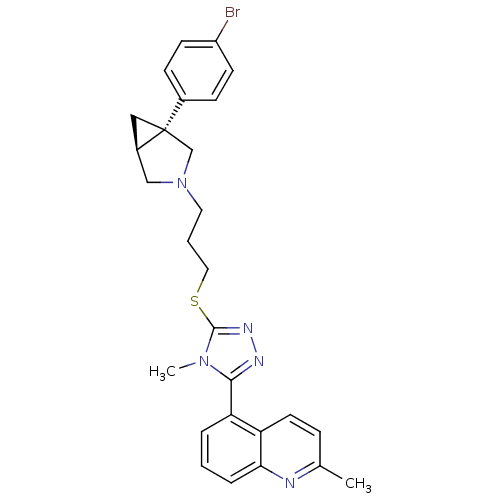
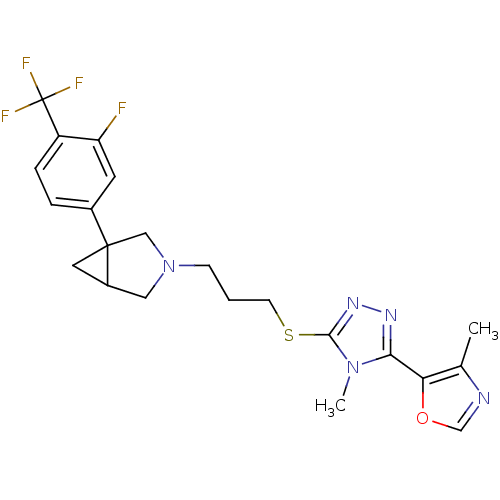
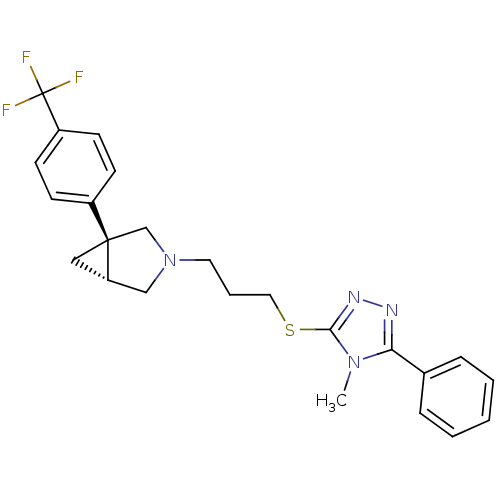
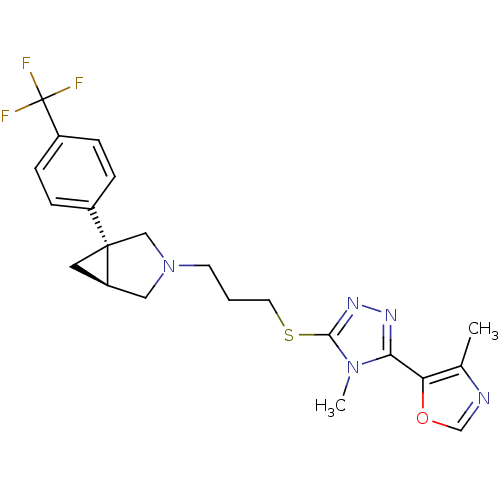
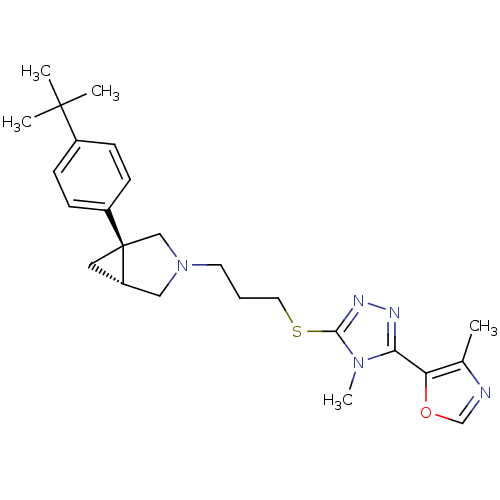
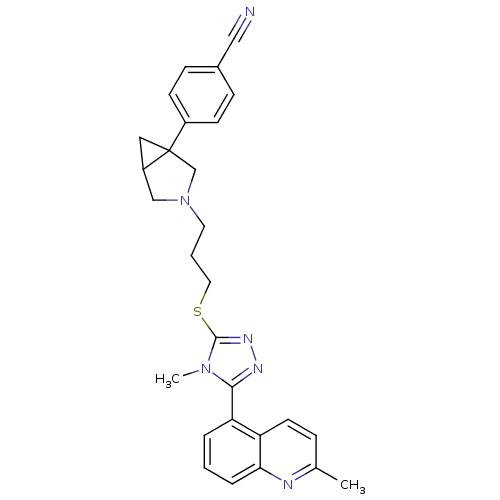
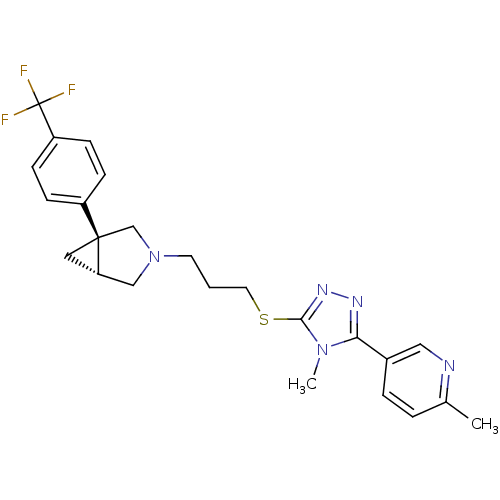
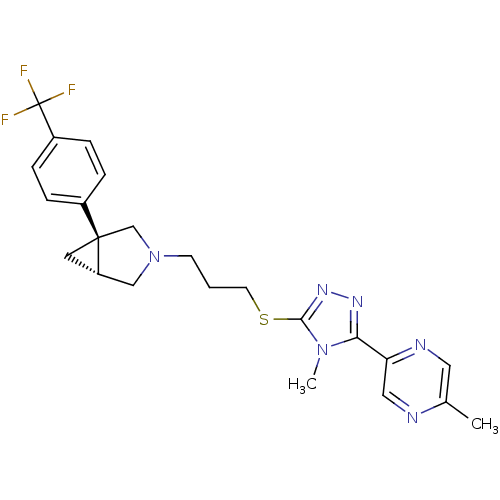
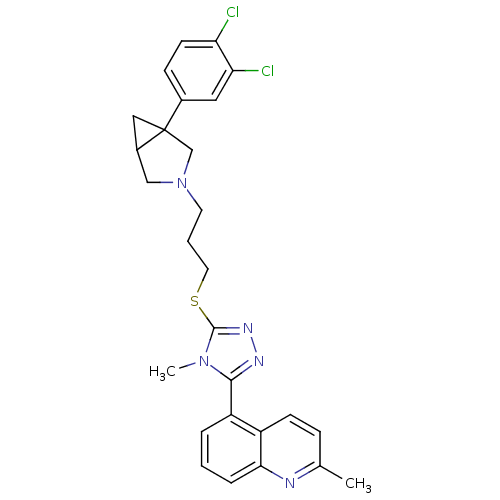
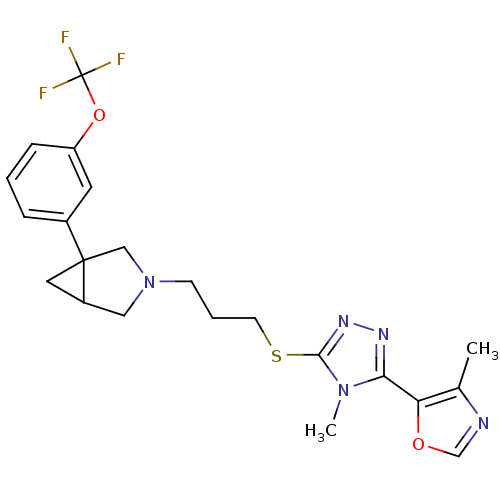
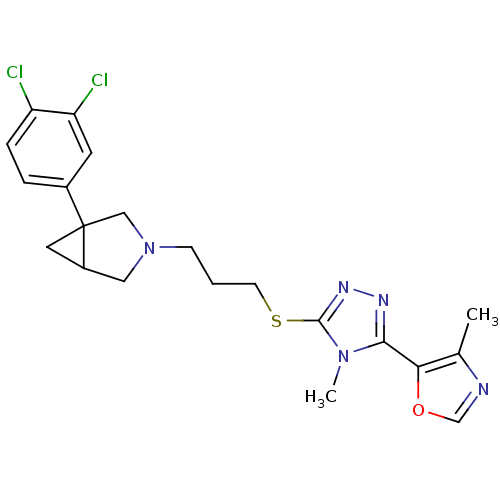
Report error Found 131 Enz. Inhib. hit(s) with all data for entry = 50031375
Affinity DataIC50: 110nMAssay Description:Inhibition of human ERG by patch clamp electrophysiology assayMore data for this Ligand-Target Pair
Affinity DataIC50: 126nMAssay Description:Displacement of [3H]dofetidile from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
Affinity DataIC50: 158nMAssay Description:Displacement of [3H]dofetidile from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
Affinity DataIC50: 200nMAssay Description:Inhibition of human ERG by patch clamp electrophysiology assayMore data for this Ligand-Target Pair
Affinity DataIC50: 200nMAssay Description:Displacement of [3H]dofetidile from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
Affinity DataIC50: 251nMAssay Description:Displacement of [3H]dofetidile from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
Affinity DataIC50: 251nMAssay Description:Displacement of [3H]dofetidile from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
Affinity DataIC50: 251nMAssay Description:Displacement of [3H]dofetidile from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
Affinity DataIC50: 251nMAssay Description:Displacement of [3H]dofetidile from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
Affinity DataIC50: 251nMAssay Description:Displacement of [3H]dofetidile from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
Affinity DataIC50: 316nMAssay Description:Displacement of [3H]dofetidile from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
Affinity DataIC50: 316nMAssay Description:Displacement of [3H]dofetidile from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
Affinity DataIC50: 350nMAssay Description:Inhibition of human ERG by patch clamp electrophysiology assayMore data for this Ligand-Target Pair
Affinity DataIC50: 390nMAssay Description:Inhibition of human ERG by patch clamp electrophysiology assayMore data for this Ligand-Target Pair
Affinity DataIC50: 398nMAssay Description:Displacement of [3H]dofetidile from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
Affinity DataIC50: 398nMAssay Description:Displacement of [3H]dofetidile from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
Affinity DataIC50: 460nMAssay Description:Inhibition of human ERG by patch clamp electrophysiology assayMore data for this Ligand-Target Pair
Affinity DataIC50: 470nMAssay Description:Inhibition of human ERG by patch clamp electrophysiology assayMore data for this Ligand-Target Pair
Affinity DataIC50: 500nMAssay Description:Inhibition of human CYP1A2More data for this Ligand-Target Pair
Affinity DataIC50: 501nMAssay Description:Displacement of [3H]dofetidile from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
Affinity DataIC50: 501nMAssay Description:Displacement of [3H]dofetidile from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
Affinity DataIC50: 501nMAssay Description:Displacement of [3H]dofetidile from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
Affinity DataIC50: 501nMAssay Description:Displacement of [3H]dofetidile from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
Affinity DataIC50: 630nMAssay Description:Displacement of [3H]dofetidile from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
Affinity DataIC50: 631nMAssay Description:Displacement of [3H]dofetidile from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
Affinity DataIC50: 631nMAssay Description:Displacement of [3H]dofetidile from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
Affinity DataIC50: 631nMAssay Description:Displacement of [3H]dofetidile from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
Affinity DataIC50: 631nMAssay Description:Displacement of [3H]dofetidile from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
Affinity DataIC50: 631nMAssay Description:Displacement of [3H]dofetidile from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
Affinity DataIC50: 631nMAssay Description:Displacement of [3H]dofetidile from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
Affinity DataIC50: 794nMAssay Description:Displacement of [3H]dofetidile from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
Affinity DataIC50: 794nMAssay Description:Displacement of [3H]dofetidile from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
Affinity DataIC50: 794nMAssay Description:Displacement of [3H]dofetidile from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
Affinity DataIC50: 794nMAssay Description:Displacement of [3H]dofetidile from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
Affinity DataIC50: 794nMAssay Description:Displacement of [3H]dofetidile from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
Affinity DataIC50: 794nMAssay Description:Displacement of [3H]dofetidile from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
Affinity DataIC50: 794nMAssay Description:Displacement of [3H]dofetidile from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
Affinity DataIC50: 794nMAssay Description:Displacement of [3H]dofetidile from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
Affinity DataIC50: 794nMAssay Description:Displacement of [3H]dofetidile from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
Affinity DataIC50: 999nMAssay Description:Displacement of [3H]dofetidile from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
Affinity DataIC50: 999nMAssay Description:Displacement of [3H]dofetidile from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
Affinity DataIC50: 999nMAssay Description:Displacement of [3H]dofetidile from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
Affinity DataIC50: 999nMAssay Description:Displacement of [3H]dofetidile from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
Affinity DataIC50: 999nMAssay Description:Displacement of [3H]dofetidile from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+3nMAssay Description:Displacement of [3H]dofetidile from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+3nMAssay Description:Displacement of [3H]dofetidile from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+3nMAssay Description:Displacement of [3H]dofetidile from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+3nMAssay Description:Displacement of [3H]dofetidile from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+3nMAssay Description:Displacement of [3H]dofetidile from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+3nMAssay Description:Displacement of [3H]dofetidile from human ERG by scintillation proximity assayMore data for this Ligand-Target Pair