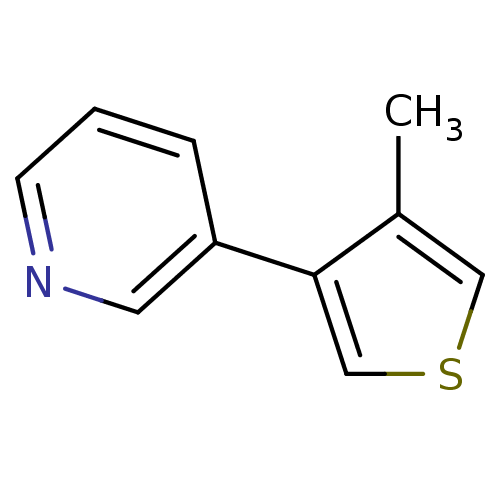
BDBM12358 3-(4-methylthiophen-3-yl)pyridine::CHEMBL179704::US8609708, 17::US8609708,17::nicotine 3-heteroaromatic analogue 11
SMILES Cc1cscc1-c1cccnc1
InChI Key InChIKey=RRFCTHIOMKUJRQ-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 13 hits for monomerid = 12358
Found 13 hits for monomerid = 12358
Affinity DataKi: 250nMAssay Description:Effect on coumarin 7-hydroxylation by human Cytochrome P-450 2A6More data for this Ligand-Target Pair
Affinity DataKi: 900nMAssay Description:To measure CYP2A6 activity, coumarin 7-hydroxylation was determined. The formation of the coumarin metabolite, 7-hydroxycoumarin, was determined fluo...More data for this Ligand-Target Pair
Affinity DataIC50: 1.50E+3nMAssay Description:Inhibitory concentration value against human cytochrome P-450 2A6More data for this Ligand-Target Pair
Affinity DataIC50: 1.85E+3nMAssay Description:The inhibition of human CYP2A6-mediated 7-hydroxy coumarin formation was evaluated in the presence of 95 selected test compounds in a standard assay ...More data for this Ligand-Target Pair
Affinity DataIC50: 1.85E+3nMAssay Description:The inhibition of human CYP2A6-mediated 7-hydroxy coumarin formation was evaluated in the presence of 95 selected test compounds in a standard assay ...More data for this Ligand-Target Pair
Affinity DataIC50: 6.30E+3nMAssay Description:Inhibitory concentration value against human cytochrome P-450 2E1More data for this Ligand-Target Pair
Affinity DataIC50: 6.30E+3nMAssay Description:Inhibitory concentration value against human cytochrome P-450 2B6More data for this Ligand-Target Pair
Affinity DataIC50: 2.49E+4nMAssay Description:Inhibitory concentration value against human cytochrome P-450 2C19More data for this Ligand-Target Pair
Affinity DataIC50: 2.50E+4nMAssay Description:To gain insight into the selectivity of the synthetic compounds, nicotine, nicotine related alkaloids and nicotine metabolites for inhibition of othe...More data for this Ligand-Target Pair
Affinity DataKi: <2.50E+4nMAssay Description:To measure CYP2A6 activity, coumarin 7-hydroxylation was determined. The formation of the coumarin metabolite, 7-hydroxycoumarin, was determined fluo...More data for this Ligand-Target Pair
Affinity DataIC50: 8.09E+4nMAssay Description:Inhibitory concentration value against human cytochrome P-450 3A4More data for this Ligand-Target Pair
Affinity DataIC50: 1.23E+5nMAssay Description:Inhibitory concentration against human cytochrome P-450 2C9More data for this Ligand-Target Pair
Affinity DataIC50: 1.93E+5nMAssay Description:Inhibitory concentration value against human cytochrome P-450 2D6More data for this Ligand-Target Pair