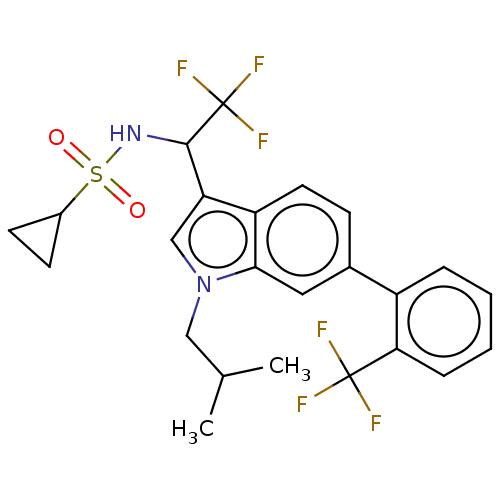
BDBM50630588 CHEMBL5432192
SMILES CC(C)Cn1cc(C(NS(=O)(=O)C2CC2)C(F)(F)F)c2ccc(cc12)-c1ccccc1C(F)(F)F
InChI Key InChIKey=NECDAWXLCJDWTH-UHFFFAOYSA-N
Data 2 EC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 2 hits for monomerid = 50630588
Found 2 hits for monomerid = 50630588
TargetNuclear receptor subfamily 1 group D member 1(Human)
The Scripps Research Institute
Curated by ChEMBL
The Scripps Research Institute
Curated by ChEMBL
Affinity DataEC50: 110nMAssay Description:Agonist activity at his6-tagged human REV-ERBalpha LBD (281 to 614 residues) expressed in Escherichia coli assessed as increase in SMRT corepressor p...More data for this Ligand-Target Pair
TargetNuclear receptor subfamily 1 group D member 1(Human)
The Scripps Research Institute
Curated by ChEMBL
The Scripps Research Institute
Curated by ChEMBL
Affinity DataEC50: 500nMAssay Description:Agonist activity at REV-ERBalpha LBD in HEK293 cells co-transfected with pG5-UAS pCMV-Gal4 incubated for 18 hrs by multimode plate reader analysisMore data for this Ligand-Target Pair