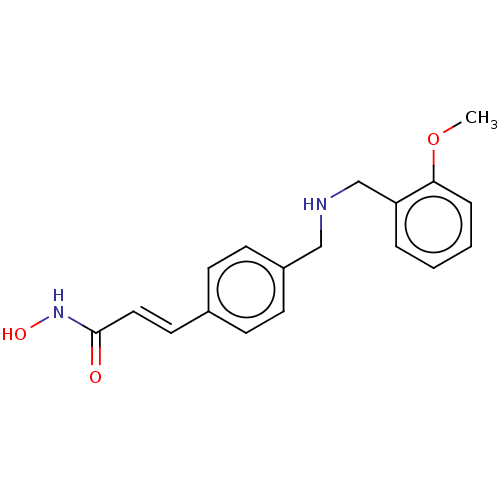
BDBM50522148 CHEMBL4454331
SMILES COc1ccccc1CNCc1ccc(\C=C\C(=O)NO)cc1
InChI Key InChIKey=SCHKQNOMUHQIJH-UHFFFAOYSA-N
Data 3 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 3 hits for monomerid = 50522148
Found 3 hits for monomerid = 50522148
Affinity DataIC50: 278nMAssay Description:Inhibition Class 1 histone deacetylase in human HeLa nuclear extracts using Fluor-de- Lys-green substrate by fluorescence assayMore data for this Ligand-Target Pair
Affinity DataIC50: 610nMAssay Description:Inhibition of human recombinant HDAC1 pre-incubated for 30 mins before substrate addition and measured after 30 mins by fluorescence based assayMore data for this Ligand-Target Pair
Affinity DataIC50: 1.49E+3nMAssay Description:Inhibition of human recombinant HDAC2 pre-incubated for 30 mins before substrate addition and measured after 30 mins by fluorescence based assayMore data for this Ligand-Target Pair