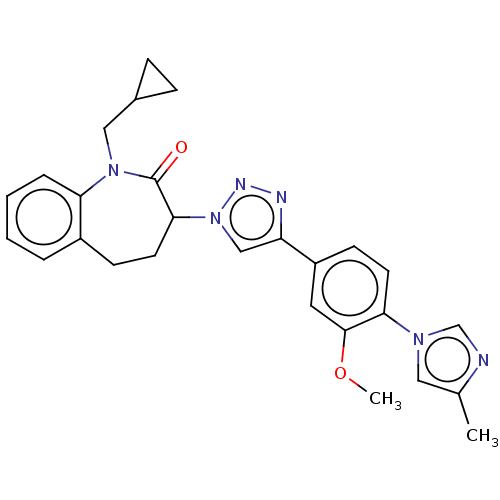
BDBM50498593 CHEMBL3609627
SMILES COc1cc(ccc1-n1cnc(C)c1)-c1cn(nn1)C1CCc2ccccc2N(CC2CC2)C1=O
InChI Key InChIKey=BIACFOILHHDYPR-UHFFFAOYSA-N
Data 3 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 3 hits for monomerid = 50498593
Found 3 hits for monomerid = 50498593
Affinity DataIC50: 370nMAssay Description:Inhibition of gamma-secretase in human SH-SY5Y cells expressing beta-APP C-terminal fragment SPA4CT assessed as decrease of amyloid beta-42 level by ...More data for this Ligand-Target Pair
TargetPotassium voltage-gated channel subfamily H member 2(Human)
Merck Research Laboratories Boston
Curated by ChEMBL
Merck Research Laboratories Boston
Curated by ChEMBL
Affinity DataIC50: 1.58E+3nMAssay Description:Binding affinity to human ERG expressed in HEK cells by MK-499 radioligand displacement assayMore data for this Ligand-Target Pair
Affinity DataIC50: 3.98E+3nMAssay Description:Inhibition of gamma-secretase in human SH-SY5Y cells expressing beta-APP C-terminal fragment SPA4CT assessed as decrease of amyloid beta-40 level by ...More data for this Ligand-Target Pair