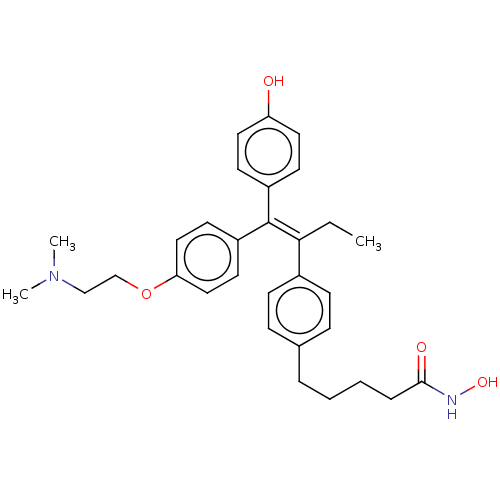
BDBM50463468 CHEMBL4237922
SMILES CC\C(=C(/c1ccc(O)cc1)c1ccc(OCCN(C)C)cc1)c1ccc(CCCCC(=O)NO)cc1
InChI Key InChIKey=YXJPLEKIYDOIFE-UHFFFAOYSA-N
Data 3 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 3 hits for monomerid = 50463468
Found 3 hits for monomerid = 50463468
Affinity DataIC50: 300nMAssay Description:Inhibition of full length recombinant human N-terminal GST-tagged HDAC6 expressed in Sf9 cells using Ac-Leu-Gly-Lys(Ac)-AMC as substrate measured aft...More data for this Ligand-Target Pair
TargetHistone deacetylase 3/Nuclear receptor corepressor 2 (HDAC3/NCoR2)(Human)
Mcgill University
Curated by ChEMBL
Mcgill University
Curated by ChEMBL
Affinity DataIC50: 734nMAssay Description:Inhibition of human full length C-terminal His-tagged HDAC3/N-terminal GST-tagged human NCOR2 (395 to 489 residues) expressed in baculovirus expressi...More data for this Ligand-Target Pair
Affinity DataIC50: 820nMAssay Description:Antagonist activity at recombinant human ERalpha expressed in HEK293 cells assessed as inhibition of estradiol-induced YFP-fused SRC1 coactivator rec...More data for this Ligand-Target Pair