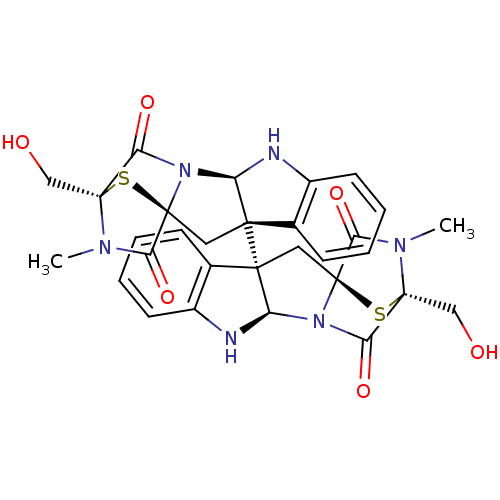
BDBM50448442 CHEMBL3124976
SMILES CN1C(=O)[C@@]23C[C@]4([C@H](Nc5ccccc45)N2C(=O)[C@]1(CO)S3)[C@]12C[C@]34S[C@](CO)(N(C)C3=O)C(=O)N4[C@H]1Nc1ccccc21
InChI Key InChIKey=GTWBRYPJUZEXNW-UHFFFAOYSA-N
Data 2 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 2 hits for monomerid = 50448442
Found 2 hits for monomerid = 50448442
Affinity DataIC50: 1.06E+4nMAssay Description:Inhibition of N-terminal GST-tagged recombinant human Histone-lysine N-methyltransferase G9a using S-(5'-adenosyl)-L-methionine chloride as substrate...More data for this Ligand-Target Pair
Affinity DataIC50: 1.07E+4nMAssay Description:Inhibition of N-terminal GST-tagged recombinant human Histone-lysine N-methyltransferase G9a using S-(5'-adenosyl)-L-methionine chloride as substrate...More data for this Ligand-Target Pair