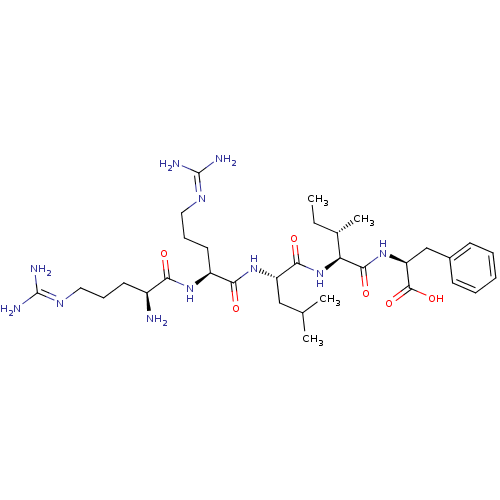
BDBM50444659 CHEMBL3098653
SMILES [#6]-[#6]-[#6@H](-[#6])-[#6@H](-[#7]-[#6](=O)-[#6@H](-[#6]-[#6](-[#6])-[#6])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#7]-[#6](=O)-[#6@@H](-[#7])-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-c1ccccc1)-[#6](-[#8])=O
InChI Key InChIKey=HIQFPEMAEIZCIP-UHFFFAOYSA-N
Data 2 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 2 hits for monomerid = 50444659
Found 2 hits for monomerid = 50444659
Affinity DataIC50: 1.61E+4nMAssay Description:Inhibition of human recombinant CDK2/cyclin A using fluoresceinyl-Ahx-Pro-Val-Lys-Arg-Arg-Leu-Phe-Gly as substrate after 45 mins by fluorescence pola...More data for this Ligand-Target Pair
TargetCyclin-dependent kinase 4/G1/S-specific cyclin-D1(Human)
University of South Carolina
Curated by ChEMBL
University of South Carolina
Curated by ChEMBL
Affinity DataIC50: 1.61E+4nMAssay Description:Inhibition of human recombinant CDK4/cyclin D1 using fluoresceinyl-Ahx-Pro-Val-Lys-Arg-Arg-Leu-(3ClPhe)-Gly as substrate after 45 mins by fluorescenc...More data for this Ligand-Target Pair