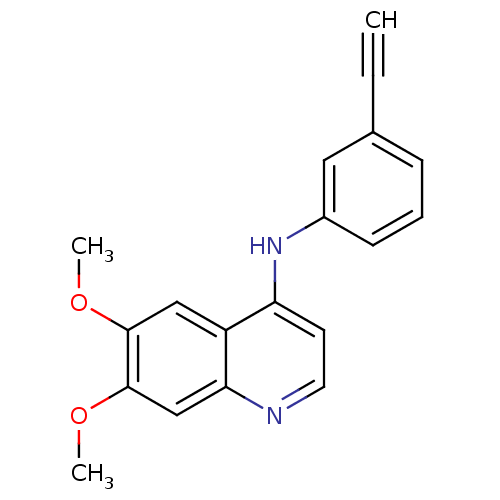
BDBM50314989 CHEMBL1092250::N-(3-Ethynylphenyl)-6,7-dimethoxyquinolin-4-amine
SMILES COc1cc2nccc(Nc3cccc(c3)C#C)c2cc1OC
InChI Key InChIKey=WMAOMQUWVSXRPR-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 5 hits for monomerid = 50314989
Found 5 hits for monomerid = 50314989
TargetEpidermal growth factor receptor(Human)
University of North Carolina at Chapel Hill
Curated by ChEMBL
University of North Carolina at Chapel Hill
Curated by ChEMBL
Affinity DataKd: 0.270nMAssay Description:Binding affinity to wild-type human partial length EGFR (R669 to V1011 residues) expressed in bacterial expression system by Kinomescan methodMore data for this Ligand-Target Pair
Affinity DataKd: 7.60nMAssay Description:Binding affinity to wild-type human partial length GAK (G13 to Y338 residues) expressed in bacterial expression system by Kinomescan methodMore data for this Ligand-Target Pair
TargetEpidermal growth factor receptor(Human)
University of North Carolina at Chapel Hill
Curated by ChEMBL
University of North Carolina at Chapel Hill
Curated by ChEMBL
Affinity DataIC50: 19nMAssay Description:Inhibition of wild type EGFR by HTRF assayMore data for this Ligand-Target Pair
TargetEpidermal growth factor receptor(Human)
University of North Carolina at Chapel Hill
Curated by ChEMBL
University of North Carolina at Chapel Hill
Curated by ChEMBL
Affinity DataIC50: 42nMAssay Description:Inhibition of EGFR in human A431 cells assessed as reduction in EGF-stimulated EGFR autophosphorylation preincuabted for 90 mins followed by EGF-stim...More data for this Ligand-Target Pair
Affinity DataIC50: 500nMAssay Description:Inhibition of tracer 5 binding to human N-terminal nano luciferase-fused GAK expressed in HEK293 cells measured after 2 hrs by nanoBRET assayMore data for this Ligand-Target Pair