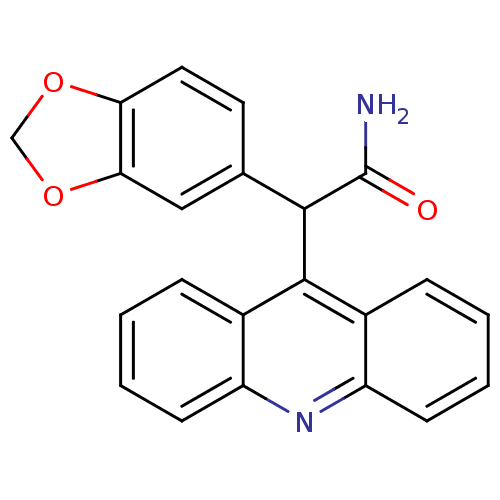
BDBM50288536 2-Acridin-9-yl-2-benzo[1,3]dioxol-5-yl-acetamide::CHEMBL100120
SMILES NC(=O)C(c1ccc2OCOc2c1)c1c2ccccc2nc2ccccc12
InChI Key InChIKey=WGZNLQUCFSXCGZ-UHFFFAOYSA-N
Data 3 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 3 hits for monomerid = 50288536
Found 3 hits for monomerid = 50288536
Affinity DataIC50: 7.90E+3nMAssay Description:Inhibition of binding of [125I]ET1 to cloned human Endothelin A receptor expressed in LtK- cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 8.50E+3nMAssay Description:Compound was tested for its ability to inhibit Endothelin A receptor induced arachidonic acid release(AARA) in rabbit renal artery vascular smooth mu...More data for this Ligand-Target Pair
Affinity DataIC50: 7.70E+4nMAssay Description:Compound was tested for its ability to inhibit Endothelin A receptor induced arachidonic acid release(AARA) in rabbit renal artery vascular smooth mu...More data for this Ligand-Target Pair