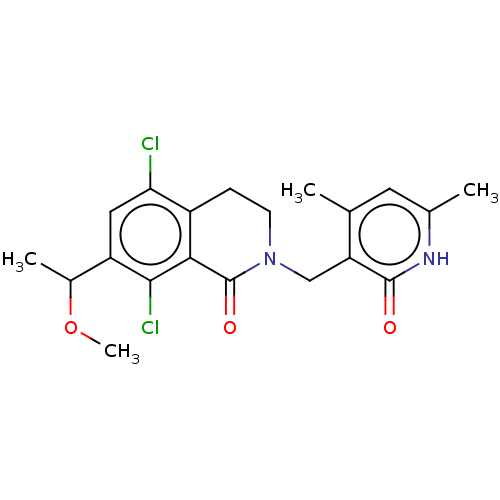
BDBM50246943 CHEMBL4081551::US10570121, Example 44
SMILES COC(C)c1cc(Cl)c2CCN(Cc3c(C)cc(C)[nH]c3=O)C(=O)c2c1Cl
InChI Key InChIKey=GZKNPZYWFSQGAJ-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 4 hits for monomerid = 50246943
Found 4 hits for monomerid = 50246943
Affinity DataKi: 22nMAssay Description:A. Compound preparation1. Prepare 10 mM stock solutions in 100% DMSO from solid material2. Serial dilute 10 mM compound stocks either 2 or 3-fold in ...More data for this Ligand-Target Pair
Affinity DataIC50: 144nMAssay Description:A. Compound preparation1. Prepare 10 mM stock solutions in 100% DMSO from solid material2. Serial dilute 10 mM compound stocks either 2 or 3-fold in ...More data for this Ligand-Target Pair
Affinity DataIC50: 1.30E+3nMAssay Description:Inhibition of EZH2 in human KARPAS422 cells assessed as reduction in H3K27me3 level after 72 hrs by ELISAMore data for this Ligand-Target Pair
Affinity DataIC50: 1.31E+3nMAssay Description:Inhibition of EZH2 in human KARPAS422 cells assessed as reduction in H3K27me3 level after 72 hrs by ELISAMore data for this Ligand-Target Pair