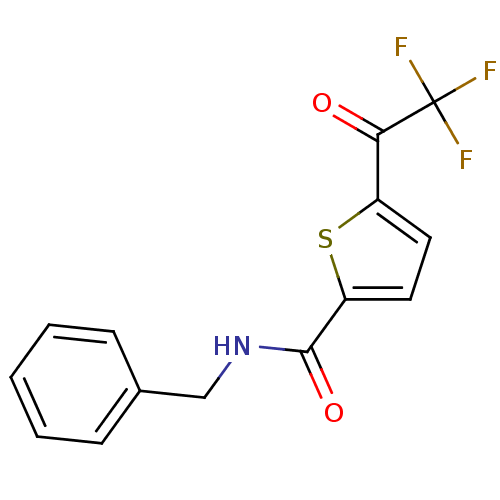
BDBM50246470 CHEMBL455455::N-benzyl-5-(2,2,2-trifluoroacetyl)thiophene-2-carboxamide
SMILES FC(F)(F)C(=O)c1ccc(s1)C(=O)NCc1ccccc1
InChI Key InChIKey=MAHDYMLFMJRXOZ-UHFFFAOYSA-N
Data 4 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 4 hits for monomerid = 50246470
Found 4 hits for monomerid = 50246470
Affinity DataIC50: 243nMAssay Description:Inhibition of His-tagged HDAC4 catalytic domain expressed in Escherichia coliMore data for this Ligand-Target Pair