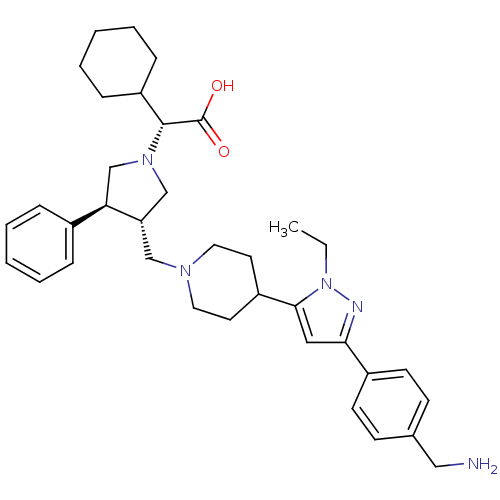
BDBM50141867 (3-{4-[5-(4-Aminomethyl-phenyl)-2-ethyl-2H-pyrazol-3-yl]-piperidin-1-ylmethyl}-4-phenyl-pyrrolidin-1-yl)-cyclohexyl-acetic acid::(R)-2-((3S,4S)-3-((4-(3-(4-(aminomethyl)phenyl)-1-ethyl-1H-pyrazol-5-yl)piperidin-1-yl)methyl)-4-phenylpyrrolidin-1-yl)-2-cyclohexylacetic acid::CHEMBL368757
SMILES CCn1nc(cc1C1CCN(C[C@H]2CN(C[C@@H]2c2ccccc2)[C@H](C2CCCCC2)C(O)=O)CC1)-c1ccc(CN)cc1
InChI Key InChIKey=YJMFAXOJBXHJLD-UHFFFAOYSA-N
Data 2 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 2 hits for monomerid = 50141867
Found 2 hits for monomerid = 50141867
Affinity DataIC50: 112nMAssay Description:Inhibitory activity against human CC chemokine receptor 5More data for this Ligand-Target Pair
Affinity DataIC50: 122nMAssay Description:Displacement of [125I]MIP1alpha from human CCR5 expressed in CHO cellsMore data for this Ligand-Target Pair