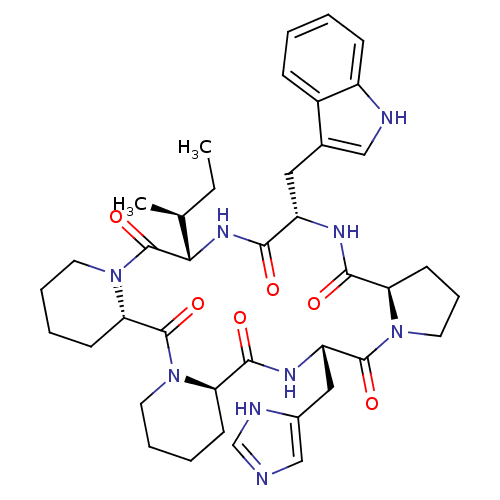
BDBM50013634 24-(1H-5-imidazolylmethyl)-16-(1H-3-indolylmethyl)-13-[1-methyl-(1S)-propyl]perhydrodipyrido[1,2-a:1,2-d]pyrrolo[1,2-j][1,4,7,10,13,16]hexaazacyclooctadecine-6,12,15,18,23,26-hexaone::CHEMBL52176
SMILES CC[C@H](C)[C@@H]1NC(=O)[C@H](Cc2c[nH]c3ccccc23)NC(=O)[C@H]2CCCN2C(=O)[C@H](Cc2cnc[nH]2)NC(=O)[C@H]2CCCCN2C(=O)[C@@H]2CCCCN2C1=O
InChI Key InChIKey=WCNGAVKBHXZVJO-UHFFFAOYSA-N
Data 3 KI
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 3 hits for monomerid = 50013634
Found 3 hits for monomerid = 50013634
Affinity DataKi: 7.80nMAssay Description:Inhibition of [3H]- oxytocin binding to rat uterine Oxytocin receptorMore data for this Ligand-Target Pair
Affinity DataKi: 1.80E+3nMAssay Description:Inhibition of [3H]arginine vasopressin binding to rat kidney medulla Vasopressin V2 receptorMore data for this Ligand-Target Pair
Affinity DataKi: 2.20E+3nMAssay Description:Inhibition of [3H]arginine vasopressin binding to rat liver Vasopressin V1 receptorMore data for this Ligand-Target Pair