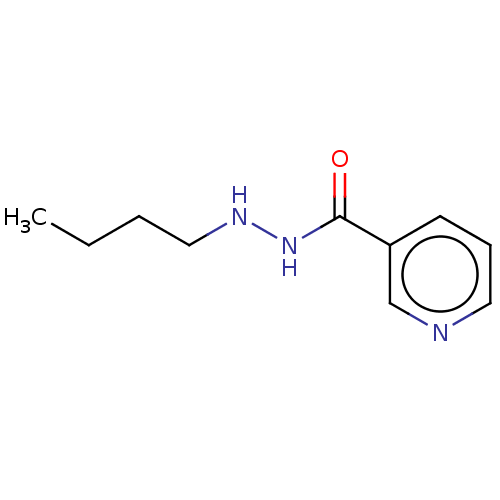
BDBM468597 US10807944, Compound RLS2-289::US10870618, Compound 1d::US11731934, Compound RLS2-289
SMILES CCCCNNC(=O)c1cccnc1
InChI Key InChIKey=NBKPFBBNYBOQPN-UHFFFAOYSA-N
Data 9 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 9 hits for monomerid = 468597
Found 9 hits for monomerid = 468597
Affinity DataIC50: 1.55E+3nMAssay Description:Recombinant HDACs 1, 2, and 3 (BPS Biosciences) were diluted to a concentration of 1 nM in HDAC buffer. 10 uL of this solution was added in 96-well f...More data for this Ligand-Target Pair
Affinity DataIC50: 1.55E+3nMAssay Description:Allosteric inhibition of C-terminal His-tagged recombinant human HDAC3 expressed in Sf9 cells pre-incubated for 2 hrs before acetylated lysine-aminom...More data for this Ligand-Target Pair
Affinity DataIC50: 4.35E+3nMAssay Description:These SAR data indicate that a tripartite structure of this scaffold with a central C(O) NH NH unit flanked by a phenyl group and a short aliphatic c...More data for this Ligand-Target Pair
Affinity DataIC50: 4.68E+3nMAssay Description:Allosteric inhibition of HDAC3 in HEK293 cell lysates pre-incubated for 2 hrs before acetylated lysine-aminomethyl coumarin-BOC addition and measured...More data for this Ligand-Target Pair
Affinity DataIC50: 1.45E+4nMAssay Description:These SAR data indicate that a tripartite structure of this scaffold with a central C(O) NH NH unit flanked by a phenyl group and a short aliphatic c...More data for this Ligand-Target Pair
Affinity DataIC50: 1.45E+4nMAssay Description:These SAR data indicate that a tripartite structure of this scaffold with a central C(O) NH NH unit flanked by a phenyl group and a short aliphatic c...More data for this Ligand-Target Pair
Affinity DataIC50: 5.67E+4nMAssay Description:These SAR data indicate that a tripartite structure of this scaffold with a central C(O) NH NH unit flanked by a phenyl group and a short aliphatic c...More data for this Ligand-Target Pair